A) \[PhCOC{{H}_{2}}C{{H}_{3}}\]
B) \[PhC{{H}_{2}}C{{H}_{2}}CHO\]
C) \[PhCOCOMe\]
D) \[PhC{{H}_{2}}COC{{H}_{3}}\]
Correct Answer: A
Solution :
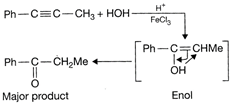 Due to higher stability of this cation \[[Ph-\overset{\oplus }{\mathop{C}}\,=CMe]\]than methyl cation, the\[-O{{H}^{o-}}\] will attack on the benzylic cation. \[\therefore \]\[PhCOC{{H}_{2}}C{{H}_{3}}\] would be the major product. Hence, the correct option is [a].
Due to higher stability of this cation \[[Ph-\overset{\oplus }{\mathop{C}}\,=CMe]\]than methyl cation, the\[-O{{H}^{o-}}\] will attack on the benzylic cation. \[\therefore \]\[PhCOC{{H}_{2}}C{{H}_{3}}\] would be the major product. Hence, the correct option is [a].
You need to login to perform this action.
You will be redirected in
3 sec
