[a]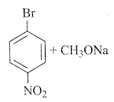 | [b]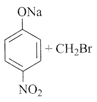 |
A) A
B) B
C) Both [a] and [b]
D) None of these
Correct Answer: C
Solution :
Even though when aromatic halides cannot be used in Williamson ether synthesis, yet because of its strong electron withdrawing group like \[N{{O}_{2}}\]at ortho and para positions that are present it is predicted that synthesis may occur. For e.g.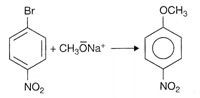
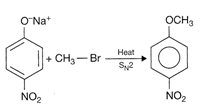 Hence, the correct option is [c].
Hence, the correct option is [c].
You need to login to perform this action.
You will be redirected in
3 sec
