A) High oxidation state of phosphorus
B) Presence of two -OH groups and one P-H bond
C) Presence of one -OH group and two P-H bonds
D) High electron gain enthalpy of phosphorus
Correct Answer: C
Solution :
Strong reducing behaviour of \[{{H}_{3}}P{{O}_{2}}\] All oxy-acid of phosphorus which contain P-H bond act as reductant.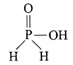 Presence of one -OH group and two P-H bonds.
Presence of one -OH group and two P-H bonds.
You need to login to perform this action.
You will be redirected in
3 sec
