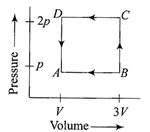
A) PV
B) 2 PV
C) 4 PV
D) \[{\scriptstyle{}^{1}/{}_{2}}PV\]
Correct Answer: B
Solution :
From the first law of thermodynamics \[\Delta Q=\Delta U+\Delta W\] For cyclic process \[\Delta U=0\] Hence Q = W Work done W= area under P-V diagram \[\Rightarrow \]\[Q=-2PV\]You need to login to perform this action.
You will be redirected in
3 sec
