A)
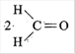
B) \[2C{{O}_{2}}\]
C) 2 HCOOH
D)
![]()
Correct Answer: A
Solution :
\[\underset{C{{H}_{2}}OH}{\mathop{\underset{|}{\mathop{C{{H}_{2}}OH}}\,}}\,\xrightarrow{HI{{O}_{4}}}2HCHO\] \[HI{{O}_{4}}\]oxidises-\[C{{H}_{2}}OH\]to HCHO and breaks the C-C bond of terminal \[C{{H}_{2}}OH\]groups.You need to login to perform this action.
You will be redirected in
3 sec
