A) \[{{H}_{3}}{{O}^{+}}\]
B) \[BF_{4}^{-}\]
C) \[HF_{2}^{-}\]
D) \[NH_{4}^{+}\]
Correct Answer: C
Solution :
Only \[HF_{2}^{-}\] has H-bonding \[[F-H-{{F}^{-}}]\] rest all the molecules have co-ordinate bonds;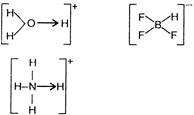
You need to login to perform this action.
You will be redirected in
3 sec
