A) \[{{[Ni{{(CN)}_{4}}]}^{2-}}\]
B) \[{{[Pd{{(CN)}_{4}}]}^{2-}}\]
C) \[{{[PdC{{l}_{4}}]}^{2-}}\]
D) \[{{[NiC{{l}_{4}}]}^{2-}}\]
Correct Answer: D
Solution :
In \[{{[NiC{{l}_{4}}]}^{2-}}\]oxidation state of \[Ni\] is \[+2\] \[N{{i}^{2+}}=1{{s}^{2}},2{{s}^{2}},2{{p}^{6}},3{{s}^{2}}3{{p}^{6}}3{{d}^{8}}\]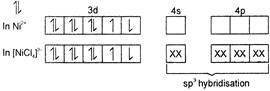 Hence, due to \[S{{P}^{3}}\] hybridisation of \[N{{i}^{2+}}\] in \[{{[NiC{{l}_{4}}]}^{2}},\]the shape of \[{{[NiC{{l}_{4}}]}^{2-}}\] is tetrahedral.
Hence, due to \[S{{P}^{3}}\] hybridisation of \[N{{i}^{2+}}\] in \[{{[NiC{{l}_{4}}]}^{2}},\]the shape of \[{{[NiC{{l}_{4}}]}^{2-}}\] is tetrahedral.
You need to login to perform this action.
You will be redirected in
3 sec
