A) All oxides of alkali metals except \[{{C}_{S2}}O\]possess anti-fluorite structure
B) The crystal structures of \[NaHC{{O}_{3}}\]and \[KHC{{O}_{3}}\]are similar
C) \[L{{i}_{2}}C{{O}_{3}},\]\[L{{i}_{3}}P{{O}_{4}}\] and LiF are insoluble in water
D) None of these
Correct Answer: B
Solution :
The crystal structures of \[NaHC{{O}_{3}}\] and \[KHC{{O}_{3}}\]both show H-bonding but are different. In \[NaHC{{O}_{3}},\]\[HCO_{3}^{-}\] ions are linked into an infinite chain while in \[KHC{{O}_{3}}\]a dimeric anion is formed.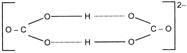
You need to login to perform this action.
You will be redirected in
3 sec
