A) \[C{{H}_{3}}COOH+C{{H}_{3}}COC{{H}_{3}}\]
B) \[C{{H}_{3}}COOH+C{{H}_{3}}C{{H}_{2}}COOH\]
C) \[C{{H}_{3}}CHO+C{{H}_{3}}C{{H}_{2}}CHO\]
D) \[C{{H}_{3}}COOH+C{{O}_{2}}\]
Correct Answer: B
Solution :
\[C{{H}_{3}}-C=C-C{{H}_{2}}-C{{H}_{3}}\xrightarrow{{{O}_{3}}}\]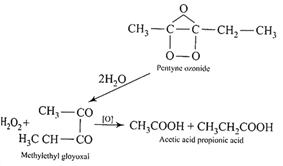 The glyoxal formed as an intermediate is oxidised by \[{{H}_{2}}{{O}_{2}}\]to give the acids.
The glyoxal formed as an intermediate is oxidised by \[{{H}_{2}}{{O}_{2}}\]to give the acids.
You need to login to perform this action.
You will be redirected in
3 sec
