A) \[\left[ Co{{(N{{H}_{3}})}_{3}}C{{l}_{3}} \right]\]
B) \[\left[ Co{{\left( en \right)}_{3}} \right]C{{l}_{3}}\]
C) \[\left[ Co{{\left( en \right)}_{2}}C{{l}_{2}} \right]Cl\]
D) \[\left[ Co\left( en \right){{\left( N{{H}_{3}} \right)}_{2}}C{{l}_{2}} \right]Cl\]
Correct Answer: A
Solution :
Because it has only two possible isomers, cis & trans as follows: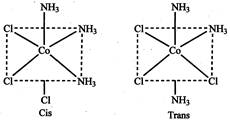 Optical isomers: Optical isomers rotate the plane of polarized light in opposite directions that is they are non-super-imposable images of each other.
Optical isomers: Optical isomers rotate the plane of polarized light in opposite directions that is they are non-super-imposable images of each other.
You need to login to perform this action.
You will be redirected in
3 sec
