A) 2T
B) 4T
C) \[\sqrt{2}T\]
D) \[2\sqrt{2}T\]
Correct Answer: C
Solution :
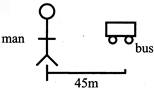 Bus stats from rest \[{{\operatorname{v}}^{2}}={{u}^{2}}+2as\] \[{{\operatorname{v}}^{2}} =0+2\times 2.5\times 45\] \[{{\operatorname{v}}^{2}} =5\times 45\] \[{{\operatorname{v}}^{2}} = 225\] \[\operatorname{v} = 15m/s\]
Bus stats from rest \[{{\operatorname{v}}^{2}}={{u}^{2}}+2as\] \[{{\operatorname{v}}^{2}} =0+2\times 2.5\times 45\] \[{{\operatorname{v}}^{2}} =5\times 45\] \[{{\operatorname{v}}^{2}} = 225\] \[\operatorname{v} = 15m/s\]
You need to login to perform this action.
You will be redirected in
3 sec
