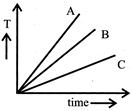
A) A
B) B
C) C
D) Equal for all
Correct Answer: A
Solution :
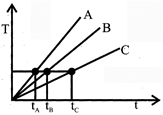 Substance having more heat capacity takes longer time to get heated to a higher temperature and longer time to get cooled. If we draw a line parallel to the time axis, then it cuts the given graphs at three different points corresponding points on the time axis show that \[{{\operatorname{t}}_{C}}> {{t}_{B}}> {{t}_{C}} or {{C}_{C}} > {{C}_{B}}> {{C}_{A}}\]
Substance having more heat capacity takes longer time to get heated to a higher temperature and longer time to get cooled. If we draw a line parallel to the time axis, then it cuts the given graphs at three different points corresponding points on the time axis show that \[{{\operatorname{t}}_{C}}> {{t}_{B}}> {{t}_{C}} or {{C}_{C}} > {{C}_{B}}> {{C}_{A}}\]
You need to login to perform this action.
You will be redirected in
3 sec
