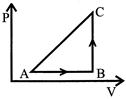
A) 10J
B) 70J
C) 84J
D) 134J
Correct Answer: D
Solution :
[d] Heat given \[\Delta Q=20cal\text{ }=\text{ }20\times 4.2=84J\] Work done \[\Delta W=-\,50\text{ }J\] [Process Anticlockwise] Ist law of thermodynamics \[\Delta U=\Delta Q-\Delta W\] \[=84-(-50)\] \[=134J\]You need to login to perform this action.
You will be redirected in
3 sec
