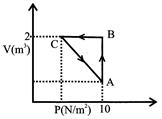
A) - 5J
B) - 10J
C) - 15J
D) - 20J
Correct Answer: A
Solution :
[a] Process is cyclic, \[\Delta \,U=0\] Total work done \[{{W}_{AB}}+{{W}_{BC}}+{{W}_{CA}}\] Ist law of thermodynamic \[{{W}_{AB}}=P\Delta V=10(2-1)=10J\] \[{{W}_{BC}}=P\Delta V=0\] \[\Delta Q=\Delta U+W\] \[5J=10+0+{{W}_{CA}}\] \[{{W}_{CA}}=-\,5J\] Note W= +ve clockwise W= -ve anticlockwise Q= +ve heat ab- sorbed Q=-ve heat releasedYou need to login to perform this action.
You will be redirected in
3 sec
