A) Boron halides are all monomeric while these of \[Al\] is dimeric
B) Boron halides and aluminium halides exist as monomeric.
C) Boron halides and aluminium halides are lewis acid
D) \[{{B}_{2}}{{O}_{3}}\] alone is acidic while \[A{{l}_{2}}{{O}_{3}}\] is amphoteric
Correct Answer: B
Solution :
All the halides of boron are volatile highly reactive and monomeric while aluminium halides exist as dimeric molecules.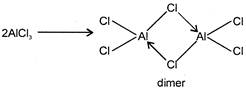
You need to login to perform this action.
You will be redirected in
3 sec
