A) Iron
B) Magnesium
C) Aluminium
D) Phosphorus
Correct Answer: D
Solution :
For n-type semiconductors, impurity atom should have 5 valence electrons. Silicon has 4 valence electrons, when phosphorous with 5 valence electrons in its outermost orbit is added, then four of the five valence electrons of the impurity atom form covalent bonds with each valence electron of four silicon atoms and fifth valence electrons becomes free to move in the crystal. As the electrons are charge carriers they are called n-type.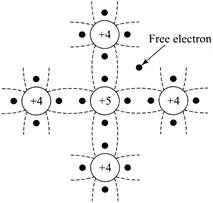
You need to login to perform this action.
You will be redirected in
3 sec
