A)
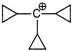
B)
![]()
C)
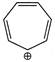
D)
![]()
Correct Answer: A
Solution :
Due to resonance of 3-propyl ring through \[\sigma -bond\] resonance has highest stability.You need to login to perform this action.
You will be redirected in
3 sec
