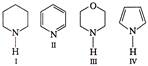
A) \[\operatorname{IV}>I>III>II\]
B) \[III>I>II>IV\]
C) \[II > I > III > IV\]
D) \[\operatorname{I} > III > II > IV\]
Correct Answer: B
Solution :
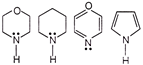 More free the electron present on N atom as a lone pair, more will be its basic strength, In IV lone pair of nitrogen is involved in acromatic 671 electrons, hence IV is least basic among all.
More free the electron present on N atom as a lone pair, more will be its basic strength, In IV lone pair of nitrogen is involved in acromatic 671 electrons, hence IV is least basic among all.
You need to login to perform this action.
You will be redirected in
3 sec
