A) ionisation isomerism, geometrical isomerism and optical isomerism
B) linkage isomerism, geometrical isomerism and optical isomerism
C) linkage isomerism. ionisation isomerism and optical isomerism
D) linkage isomerism, ionisation isomerism and geometrical isomerism
Correct Answer: D
Solution :
\[-N{{O}_{2}}\] has two types of linkage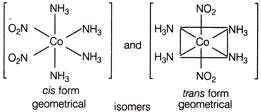 \[\left[ Co {{\left( N{{N}_{3}} \right)}_{4}} {{\left( N{{O}_{2}} \right)}_{2}} \right] Cl\] and \[\left[ CO {{\left( N{{N}_{3}} \right)}_{4}}\,NO{{ & }_{2}}Cl \right]N{{O}_{2}}\] are ionisation isomers.
\[\left[ Co {{\left( N{{N}_{3}} \right)}_{4}} {{\left( N{{O}_{2}} \right)}_{2}} \right] Cl\] and \[\left[ CO {{\left( N{{N}_{3}} \right)}_{4}}\,NO{{ & }_{2}}Cl \right]N{{O}_{2}}\] are ionisation isomers.
You need to login to perform this action.
You will be redirected in
3 sec
