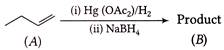
A) B is a secondary alcohol
B) B is a primary alcohol
C) B is a tertiary alcohol
D) B is an achiral molecule
Correct Answer: A
Solution :
This reaction is an example of oxymercuration demercuration reaction which proceeds as follows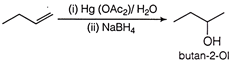 B is a secondary alcohol because OH is attached to a secondary carbon atom,
B is a secondary alcohol because OH is attached to a secondary carbon atom, You need to login to perform this action.
You will be redirected in
3 sec
