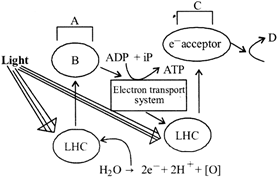
A) A - P 700 B - \[{{H}^{+}}\] acceptor C - P680 D - \[NAD{{P}^{+}}\]
B) A ? Photosystem I B - \[{{e}^{-}}\] acceptor C - Photosystem II D - \[NADP{{H}_{2}}+ATP\]
C) A ? Photosystem I B - \[{{H}^{+}}\] acceptor C - P700 D - \[NADPH\]
D) A ? Photosystem III B - \[{{e}^{-}}\] acceptor C - Photosystem I D - \[NADPH+{{H}^{+}}\]
Correct Answer: D
Solution :
[d] Given figure represents Z scheme of light reaction. The whole scheme of transfer of electrons, starting from the PS II, uphill to the acceptor, down the electron transport to PS I, excitation of electrons transfer to another acceptor, and finally down hill to \[NAD{{P}^{+}}\] causing it to be reduced to \[NADPH+{{H}^{+}}\] is called the Z scheme, due to its characteristic shape.You need to login to perform this action.
You will be redirected in
3 sec
