A) c < a < b
B) b < c < a
C) a < b < a
D) a < c < b
Correct Answer: D
Solution :
[d]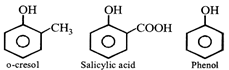 Electron releasing groups \[(-C{{H}_{3}},-OC{{H}_{3}},-NC{{H}_{3}}\,etc)\] intensify the negative charge of phenoxide ion, i.e., destablises it hence decrease ionization of parent phenol. Therefore decreases acidity while electron donating groups \[(-N{{O}_{2}},-COOH,-CHO\,etc.)\] increases acidity.
Electron releasing groups \[(-C{{H}_{3}},-OC{{H}_{3}},-NC{{H}_{3}}\,etc)\] intensify the negative charge of phenoxide ion, i.e., destablises it hence decrease ionization of parent phenol. Therefore decreases acidity while electron donating groups \[(-N{{O}_{2}},-COOH,-CHO\,etc.)\] increases acidity.
You need to login to perform this action.
You will be redirected in
3 sec
