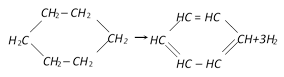Chemisorption
A)
Involves the weak attractive interactions between adsorbent and adsorbate
done
clear
B)
Is irreversible in nature
done
clear
C)
Decreases with increase of temperature
done
clear
D)
Involves multilayer formation of adsorbent on adsorbate
done
clear
View Answer play_arrow
Which one of the following statement is not correct?
A)
The extent of adsorption depends on the nature of the adsorbent and adsorbate
done
clear
B)
The extent of adsorption depends on the pressure of the gas
done
clear
C)
The extent of adsorption depends on the temperature
done
clear
D)
The extent of adsorption has no upper limit Not Available Solution Available Solution
done
clear
View Answer play_arrow
Which of the following kinds of catalysis can be explained by the adsorption theory?
A)
Homogeneous catalysis
done
clear
B)
Acid base catalysis
done
clear
C)
Heterogeneous catalysis
done
clear
D)
Enzyme catalysis
done
clear
View Answer play_arrow
In Haber's process for the manufacture of ammonia
A)
Finely divided iron is used as catalyst
done
clear
B)
Finely divided molybdenum is used as catalyst
done
clear
C)
Finely divided nickel is used as catalyst
done
clear
D)
No catalyst is necessary
done
clear
View Answer play_arrow
Which one of the following statement is wrong in case of enzyme catalysis?
A)
Enzymes work best at an optimum temperature
done
clear
B)
Enzymes work at an optimum \[pH\]
done
clear
C)
Enzymes are highly specific for substances
done
clear
D)
An enzyme raises activation energy
done
clear
View Answer play_arrow
Which of the statement is wrong among the following
A)
Haber's process of \[N{{H}_{3}}\] requires iron as catalyst
done
clear
B)
Friedel-Craft's reaction uses anhydrous \[AlC{{l}_{3}}\]
done
clear
C)
Hydrogenation of oils uses iron as catalyst
done
clear
D)
Oxidation of \[S{{O}_{2}}\] to \[S{{O}_{3}}\] requires \[{{V}_{2}}{{O}_{5}}\]
done
clear
View Answer play_arrow
Catalyst used in the oxidation of \[S{{O}_{2}}\to S{{O}_{3}}\]
A)
Nickel
done
clear
B)
\[ZnO.C{{r}_{2}}{{O}_{3}}\]
done
clear
C)
\[{{V}_{2}}{{O}_{5}}\]
done
clear
D)
Iron
done
clear
View Answer play_arrow
Ferric hydroxide sol is positively charged colloid. The coagulating power of \[NO_{3}^{-},\,SO_{4}^{2-}\] and \[PO_{4}^{3-}\] ions would be in the order
A)
\[NO_{3}^{-}>SO_{4}^{2-}>PO_{4}^{3-}\]
done
clear
B)
\[SO_{4}^{2-}>NO_{3}^{-}>PO_{4}^{3-}\]
done
clear
C)
\[PO_{4}^{3-}>SO_{4}^{2-}>NO_{3}^{-}\]
done
clear
D)
\[NO_{3}^{-}=SO_{4}^{2-}=PO_{4}^{3-}\]
done
clear
View Answer play_arrow
Which of the following will have highest coagulating power for \[A{{s}_{2}}{{S}_{3}}\] colloid
A)
\[PO_{4}^{-3}\]
done
clear
B)
\[SO_{4}^{-2}\]
done
clear
C)
\[N{{a}^{+}}\]
done
clear
D)
\[A{{l}^{3+}}\]
done
clear
View Answer play_arrow
Which of the following reactions leads to the formation of a substance in the colloidal state
A)
\[Cu+HgC{{l}_{2}}\to CuC{{l}_{2}}+Hg\]
done
clear
B)
\[2HN{{O}_{3}}+3{{H}_{2}}S\to 3S+4{{H}_{2}}O+2NO\]
done
clear
C)
\[2Mg+C{{O}_{2}}\to 2MgO+C\]
done
clear
D)
\[Cu+CuC{{l}_{2}}\to C{{u}_{2}}C{{l}_{2}}\]
done
clear
View Answer play_arrow
Butter is
A)
A gel
done
clear
B)
An emulsion
done
clear
C)
A sol
done
clear
D)
Not a colloid
done
clear
View Answer play_arrow
Pick out the statement which is not relevant in the discussion of colloids
A)
Sodium aluminium silicate is used in the softening of hard water
done
clear
B)
Potash alum is used in shaving rounds and as antiseptic in medicine
done
clear
C)
Artificial rain is caused by throwing electrified sand on the clouds from an aeroplane
done
clear
D)
Deltas are formed at a place where the river pours its water into the sea
done
clear
View Answer play_arrow
Milk is
A)
Dispersed fats in oil
done
clear
B)
Dispersed fats in water
done
clear
C)
Dispersed water in fats
done
clear
D)
Dispersed water in oil
done
clear
View Answer play_arrow
The decomposition of \[{{H}_{2}}{{O}_{2}}\] can be slowed down by the addition of small amount of phosphoric acid which act as
A)
Promoter
done
clear
B)
Inhibitor
done
clear
C)
Detainer
done
clear
D)
Stopper
done
clear
View Answer play_arrow
The impurities present in rain water possess .............. charge
A)
Positive
done
clear
B)
Negative
done
clear
C)
Zero
done
clear
D)
Positive and negative
done
clear
View Answer play_arrow
If gold number of A,B,C and D are 0.005, 0.05, 0.5 and 5 respectively, then which of the following will have the highest protective power
A)
A
done
clear
B)
B
done
clear
C)
C
done
clear
D)
D
done
clear
View Answer play_arrow
Fog is a colloidal solution of
A)
Solid in gas
done
clear
B)
Liquid in gas
done
clear
C)
Gas in liquid
done
clear
D)
Gas in solid
done
clear
View Answer play_arrow
Point out the wrong statement : Physical adsorption is characterised by
A)
Attraction due to weak Vander Waal's forces
done
clear
B)
Irreversible nature of adsorption
done
clear
C)
Multimolecular adsorption layers
done
clear
D)
Decrease in adsorption with increase in temperature
done
clear
View Answer play_arrow
In Freundlich adsorption, isotherm adsorption is proportional to pressure P as
A)
\[{{P}^{0}}\]
done
clear
B)
\[P\]
done
clear
C)
\[{{P}^{n}}\]
done
clear
D)
\[{{P}^{1/n}}\]
done
clear
View Answer play_arrow
The equation for Freundlich adsorption isotherm is
A)
\[\frac{x}{m}=k{{p}^{1/n}}\]
done
clear
B)
\[x=mk{{p}^{1/n}}\]
done
clear
C)
\[x/m=k{{p}^{-n}}\]
done
clear
D)
All of these
done
clear
View Answer play_arrow
In the following reaction the catalyst used is
A)
\[A{{l}_{2}}{{O}_{3}}\]
done
clear
B)
\[C{{r}_{2}}{{O}_{3}}\]
done
clear
C)
\[C{{r}_{2}}{{O}_{3}}\] and \[A{{l}_{2}}{{O}_{3}}\]
done
clear
D)
Zn dust
done
clear
View Answer play_arrow
Regarding criteria of catalysis which one of the following statements is not true
A)
The catalyst is unchanged chemically at the end of the reaction
done
clear
B)
A small quantity of catalyst is often sufficient to bring about a considerable amount of reaction
done
clear
C)
In a reversible reaction the catalyst alters the equilibrium position
done
clear
D)
The catalyst accelerates the reaction
done
clear
View Answer play_arrow
Amongst the following chemical reactions, the one representing homogeneous catalysis is
A)
\[{{N}_{2}}(g)+3{{H}_{2}}(g)\xrightarrow{Fe}2N{{H}_{3}}(g)\]
done
clear
B)
\[2S{{O}_{2}}(g)+{{O}_{2}}(g)\xrightarrow{2NO}2S{{O}_{3}}(g)+2NO(g)\]
done
clear
C)
\[CO(g)+3{{H}_{2}}(g)\xrightarrow{Ni}C{{H}_{4}}(g)+{{H}_{2}}O\]
done
clear
D)
\[2S{{O}_{2}}(g)+{{O}_{2}}(g)\xrightarrow{{{V}_{2}}{{O}_{5}}}2S{{O}_{3}}(g)\]
done
clear
View Answer play_arrow
Enzyme activity is maximum at
A)
300 K
done
clear
B)
310 K
done
clear
C)
320 K
done
clear
D)
330 K
done
clear
View Answer play_arrow
Which of the following statements is not true for a lyophobic sol
A)
It can be easily solvated
done
clear
B)
It carries charge
done
clear
C)
The coagulation of this sol is irreversible in nature
done
clear
D)
It is less stable in a solvent
done
clear
View Answer play_arrow
Milk is a colloid in which
A)
A liquid is dispersed in liquid
done
clear
B)
A solid is dispersed in liquid
done
clear
C)
A gas is dispersed in liquid
done
clear
D)
Some suger is dispersed in water
done
clear
View Answer play_arrow
Which is a natural colloidal
A)
Sodium chloride
done
clear
B)
Urea
done
clear
C)
Canesugar
done
clear
D)
Blood
done
clear
View Answer play_arrow
Which of the following pairs of ions would be expected to form precipitate when their dilute solution are mixed
A)
\[N{{a}^{+}},\,SO_{3}^{2-}\]
done
clear
B)
\[NH_{4}^{+},\,CO_{3}^{2-}\]
done
clear
C)
\[N{{a}^{+}},\,{{S}^{-2}}\]
done
clear
D)
\[F{{e}^{+3}},\,PO_{4}^{-3}\]
done
clear
View Answer play_arrow
In which of the following Tyndall effect is not observed
A)
Suspensions
done
clear
B)
Emulsions
done
clear
C)
Sugar solution
done
clear
D)
Gold sol
done
clear
View Answer play_arrow
When a strong beam of light is passed through a colloidal solution, the light will
A)
Give a rainbow
done
clear
B)
Be scattered
done
clear
C)
Be reflected
done
clear
D)
Absorbed completely
done
clear
View Answer play_arrow
Which one is an example of miceller system
A)
Soap + water
done
clear
B)
Protein + water
done
clear
C)
Rubber + benzene
done
clear
D)
\[A{{s}_{2}}{{O}_{3}}+Fe{{(OH)}_{3}}\]
done
clear
View Answer play_arrow
Which one is Freundlich's equation
A)
\[\frac{x}{m}=\log \,K+\frac{1}{n}\log \,P\]
done
clear
B)
\[\frac{x}{m}=\exp \,(-KP)\]
done
clear
C)
\[\frac{x}{m}=K{{P}^{2}}\]
done
clear
D)
\[\log \frac{x}{m}=\log K+\frac{1}{n}\log \,C\]
done
clear
View Answer play_arrow
At isoelectric point
A)
Colloidal sol becomes highly stable
done
clear
B)
Precipitation of a colloidal sol takes place
done
clear
C)
Colloidal particles becomes uncharged
done
clear
D)
Peptization can be carried out
done
clear
View Answer play_arrow
Gold sol is an electronegative sol. The amount of electrolyte required to coagulate a certain amount of gold sol is minimum in the case of
A)
\[CaC{{l}_{2}}\]
done
clear
B)
\[NaCl\]
done
clear
C)
\[AlC{{l}_{3}}\]
done
clear
D)
\[N{{a}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
The surface tension of which of the following liquid is maximum?
A)
\[{{H}_{2}}O\]
done
clear
B)
\[{{C}_{6}}{{H}_{6}}\]
done
clear
C)
\[C{{H}_{3}}OH\]
done
clear
D)
\[{{C}_{2}}{{H}_{5}}OH\]
done
clear
View Answer play_arrow