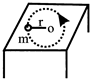
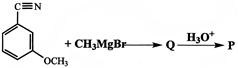Time period T of a simple pendulum may depend upon mass of the bob m; length of simple pendulum and 'g' the acceleration due to gravity. Then the value of a, b and c are:
A)
\[-\frac{1}{2},0,\frac{1}{2}\]
done
clear
B)
\[-\frac{1}{2},0,-\frac{1}{2}\]
done
clear
C)
\[0,-\frac{1}{2},\frac{1}{2}\]
done
clear
D)
\[0,\frac{1}{2},-\frac{1}{2}\]
done
clear
View Answer play_arrow
The length of the second's pendulum on the surface of earth is 1m. The length of second's pendulum on the surface of moon, where g is-tha value of earth surface, is:
A)
\[\frac{1}{6}m\]
done
clear
B)
\[6m\]
done
clear
C)
\[\frac{1}{36}m\]
done
clear
D)
\[36m\]
done
clear
View Answer play_arrow
When a particle is projected at an angle to the horizontal, it has range R and time of flight \[{{t}_{1}}\]It the same projectile is projected with the same speed at another angle to have the same range and time of flight \[{{t}_{2}}\]then,
A)
\[{{t}_{1}}+{{t}_{2}}=\frac{2R}{g}\]
done
clear
B)
\[{{t}_{1}},{{t}_{2}}=\frac{2R}{g}\]
done
clear
C)
\[{{t}_{1}}-{{t}_{2}}=\frac{2R}{g}\]
done
clear
D)
\[{{t}_{1}}\,{{t}_{2}}=\frac{2R}{g}\]
done
clear
View Answer play_arrow
L The following four wires are made of the same material. Which of these will have the largest extension. When the same tension is applied?
A)
\[\ell =1m,\,D=1mm\]
done
clear
B)
\[\ell =2m,\,D=2mm\]
done
clear
C)
\[\ell =2m,\,d=2mm\]
done
clear
D)
\[\ell =0.5m,\,D=0.5mm\]
done
clear
View Answer play_arrow
A particle of mass m is attached to M via a massless string passing through a hole O in the horizontal table as shown. Mass M is kept stationary whereas mass m is rotating in a circle of radius r with angular speed g).
A)
\[Mg=mr{{\omega }^{2}}\]
done
clear
B)
\[Mg>mr{{\omega }^{2}}\]
done
clear
C)
\[Mg<mr{{\omega }^{2}}\]
done
clear
D)
\[Mg\ge mr{{\omega }^{2}}\]
done
clear
View Answer play_arrow
If the two vectors \[\overrightarrow{A}=2\hat{i}+3\hat{j}+4\hat{k}\] and \[\overrightarrow{B}=\hat{i}+2\hat{j}-n\hat{k}\] are perpendicular, then the value of n is:
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
5
done
clear
View Answer play_arrow
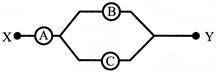
The current through the ideal diode as shown in the figure is:
A)
0 A
done
clear
B)
0.02A
done
clear
C)
0.04A
done
clear
D)
0.06A
done
clear
View Answer play_arrow
The approximate depth of an ocean is 2700m. The compressibility of water is \[45.4\times {{10}^{-11}}\text{ }P{{a}^{-1}}\] and density of water is\[{{10}^{3}}kg/{{m}^{3}}\]. What fractional compression of water will be obtained at the bottom of the ocean?
A)
\[1.4\times 10-2\]
done
clear
B)
\[0.8\times 10-2\]
done
clear
C)
\[1\times {{10}^{-2}}~\]
done
clear
D)
\[1.2\times {{10}^{-2}}\]
done
clear
View Answer play_arrow
Masses of two particles having same kinetic energies are in the ratio 1 : 2 then, their de- broglie wavelengths are in ratio:
A)
\[2:1\]
done
clear
B)
\[1:2\]
done
clear
C)
\[\sqrt{2}:1\]
done
clear
D)
\[\sqrt{3}:1\]
done
clear
View Answer play_arrow
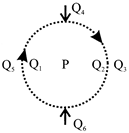
The figure shows the various positions [labelled subscripts] of small magnetized needles P and Q. The arrows show the direction of their magnetic moment. Which configuration corresponds to the lowest potential energy of all the configurations show:
A)
\[P{{Q}_{3}}\]
done
clear
B)
\[P{{Q}_{4}}\]
done
clear
C)
\[~PQ{{~}_{5}}\]
done
clear
D)
\[P{{Q}_{6}}\]
done
clear
View Answer play_arrow
The wavelength of the first line of Lyman series for hydrogen atom is equal to that of the second line of Balmer series for a hydrogen like ion. The atomic number Z of hydrogen like ion is:
A)
4
done
clear
B)
1
done
clear
C)
2
done
clear
D)
3
done
clear
View Answer play_arrow
A wire oriented in the east west direction carries a current eastward Direction of the magnetic field at a point to the south of the wire is:
A)
Vertically down
done
clear
B)
Vertically up
done
clear
C)
North east
done
clear
D)
South-East
done
clear
View Answer play_arrow
Kerosene oil rises up in the wick of a lantern because of:
A)
Diffusion of the oil through the wick
done
clear
B)
Surface tension
done
clear
C)
Buoyant force of air
done
clear
D)
The gravitational pull of the wick
done
clear
View Answer play_arrow
Electron with de Broglie wavelength\[\lambda \] fall on the target in an x-ray tube. The cut off wavelength of the emitted X-rays is:
A)
\[{{\lambda }_{o}}=\frac{2mc{{\lambda }^{2}}}{h}\]
done
clear
B)
\[{{\lambda }_{o}}=\frac{2h}{mc}\]
done
clear
C)
\[{{\lambda }_{o}}=\frac{2{{m}^{2}}{{c}^{2}}{{\lambda }^{3}}}{{{h}^{2}}}\]
done
clear
D)
\[{{\lambda }_{o}}=\lambda \]
done
clear
View Answer play_arrow
Work done by a force, \[F=\,\,(-\,6{{x}^{3}})\,\hat{i}\,N,\]in displacing a particle from \[x=4m\text{ }to\,x=-\,2m\]is:
A)
360J
done
clear
B)
240J
done
clear
C)
- 240J
done
clear
D)
- 360J
done
clear
View Answer play_arrow
The magnetic flux through a circuit of resistance R changes by an amount \[\Delta \,\phi \]in a time\[\Delta \,t\]. Then, the total quantity of electric charge Q that passes any point in the circuit during the time At is:
A)
\[Q=\frac{\Delta \phi }{R\Delta t}\]
done
clear
B)
\[Q=\frac{\Delta \phi }{R}\]
done
clear
C)
\[Q=\frac{\Delta \phi }{\Delta t}\]
done
clear
D)
\[Q=\frac{R\Delta \phi }{\Delta t}\]
done
clear
View Answer play_arrow
The given truth table is:
A B Y 0 0 0 1 0 1 0 1 1 1 1 0
A)
XOR
done
clear
B)
NAND
done
clear
C)
OR
done
clear
D)
AND
done
clear
View Answer play_arrow
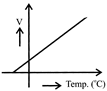
Which one of the following plots represents the variation of gravitational field on a particle with distance r due a thin spherical shell of radius R?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
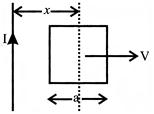
A conducting square frame of side 'a' and along straight wire carrying current I are located in the same plane a shown in the figure. The frame moves to the right with constant velocity 'v'. The emf induced in the frame will be proportional to:
A)
\[\frac{1}{(2x-a)\,(2x+a)}\]
done
clear
B)
\[\frac{1}{{{x}^{2}}}\]
done
clear
C)
\[\frac{1}{{{(2x-a)}^{2}}}\]
done
clear
D)
\[\frac{1}{{{(2x+a)}^{2}}}\]
done
clear
View Answer play_arrow
The cylindrical tube of a spray pump has radius R, one end of which has n fine holes, each of radius r. If the speed of the liquid in the tube is V, the speed of the ejection of the liquid through the holes is:
A)
\[\frac{V{{R}^{2}}}{{{n}^{3}}{{r}^{2}}}\]
done
clear
B)
\[\frac{{{V}^{2}}R}{nr}\]
done
clear
C)
\[\frac{V{{R}^{2}}}{{{n}^{2}}{{r}^{2}}}\]
done
clear
D)
\[\frac{V{{R}^{2}}}{n{{r}^{2}}}\]
done
clear
View Answer play_arrow
Two instruments having stretched strings are being played in unison. When the tension of one of the instruments is increased by 1%, 3 beats are produced in 2sec, the initial frequency of vibration of each wire is:
A)
300Hz
done
clear
B)
500Hz
done
clear
C)
100Hz
done
clear
D)
400Hz
done
clear
View Answer play_arrow
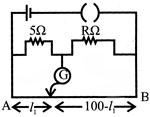
The resistances in the two arms of the meter bridge are\[5\Omega \].and\[R\Omega \], respectively. When the resistance R is shunted with an equal resistance, the new balance point is at 1.6, the resistance R is:
A)
\[10\Omega \]
done
clear
B)
\[15\Omega \]
done
clear
C)
\[20\Omega \]
done
clear
D)
\[25\Omega \]
done
clear
View Answer play_arrow
The masses of two radioactive substance are same and their half-lives are 1 hr and 2yr respectively. The ratio of their activities after 4yr will be:
A)
1 : 4
done
clear
B)
1 : 2
done
clear
C)
1 : 3
done
clear
D)
1 : 6
done
clear
View Answer play_arrow
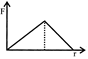
One mole of an ideal diatomic gas undergoes a transition from A to B along a path AB as shown in the figure. The change in internal energy of the gas during the transition is:
A)
- 12KJ
done
clear
B)
20KJ
done
clear
C)
- 20KJ
done
clear
D)
20J
done
clear
View Answer play_arrow
A ball is thrown vertically downwards from a height of 20m with an initial velocity\[{{V}_{{}^\circ }}\]. It collides with the ground, loses 50 percent of its energy in collision and rebounds to the same height. The initial velocity \[{{V}_{{}^\circ }}\] is:
A)
28m/s
done
clear
B)
10m/s
done
clear
C)
14m/s
done
clear
D)
20m/s
done
clear
View Answer play_arrow
A particle executes SHM with amplitude 0.2m and time period 24 sec The time require for it to move from mean position to a point 0.1 m from the mean position is:
A)
12sec
done
clear
B)
3 sec
done
clear
C)
2 sec
done
clear
D)
8 sec
done
clear
View Answer play_arrow
The current flowing through a wire depends on time as\[I=3{{t}^{2}}+2t+5\]. The charge flowing through the cross-section of the wire in time from t = 0 to t = 2s is:
A)
22C
done
clear
B)
20C
done
clear
C)
18C
done
clear
D)
5C
done
clear
View Answer play_arrow
The displacement x of a particle at the instant when its velocity is v is given by\[v=\sqrt{3x+16}\]. Its acceleration and initial velocity are.
A)
1.5 unit, 4 unit
done
clear
B)
3 unit, 4 unit
done
clear
C)
16 unit, 1.6 unit
done
clear
D)
16 unit, 3 unit
done
clear
View Answer play_arrow
A mono atomic gas at a pressure p, having a volume V expands isothermally to a volume 2V and then adiabatically to a volume 16V. The final pressure of the gas is \[\left( \gamma =\frac{5}{3} \right)\]:
A)
\[64p\]
done
clear
B)
\[32p\]
done
clear
C)
\[\frac{p}{64}\]
done
clear
D)
\[16p\]
done
clear
View Answer play_arrow
A body is projected vertically upwards. The times corresponding to height h while ascending and while descending are \[{{t}_{1}}\] and \[{{t}_{2}}\] respectively .Then, the velocity of projection is:
A)
\[g\sqrt{{{t}_{1}}{{t}_{2}}}\]
done
clear
B)
\[g\frac{{{t}_{1}}{{t}_{2}}}{{{t}_{1}}+{{t}_{2}}}\]
done
clear
C)
\[\frac{g\,\sqrt{{{t}_{1}}{{t}_{2}}}}{2}\]
done
clear
D)
\[\frac{g\,({{t}_{1}}{{t}_{2}})}{2}\]
done
clear
View Answer play_arrow
Three voltmeters A, B and C having resistances R, 1.5R and 3R, respectively are used in a circuit as shown when a potential difference is applied between X and Y, the reading of the voltmeters are and Vg respectively then:
A)
\[{{V}_{1}}={{V}_{2}}={{V}_{3}}\]
done
clear
B)
\[{{V}_{1}}<{{V}_{2}}={{V}_{3}}\]
done
clear
C)
\[{{V}_{1}}>{{V}_{2}}>{{V}_{3}}\]
done
clear
D)
\[{{V}_{1}}={{V}_{2}}={{V}_{3}}\]
done
clear
View Answer play_arrow
If the critical angle for TIR from medium to vacuum is \[30{}^\circ \], the velocity of light in medium is:
A)
\[3\times {{10}^{8}}m/s\]
done
clear
B)
\[1.5\,\times {{10}^{8}}m/s\]
done
clear
C)
\[6\,\times {{10}^{8}}m/s\]
done
clear
D)
\[\sqrt{3}\,\times {{10}^{8}}m/s\]
done
clear
View Answer play_arrow
A body takes time't' to reach the bottom of an inclined plane of angle \[\theta \] with the horizontal. If the plane is made rough, time taken new is 2t. The coefficient of friction of the rough surface is:
A)
\[\frac{3\tan \,\theta }{4}\]
done
clear
B)
\[\frac{2\tan \,\theta }{3}\]
done
clear
C)
\[\frac{\tan \,\theta }{4}\]
done
clear
D)
\[\frac{\tan \,\theta }{2}\]
done
clear
View Answer play_arrow
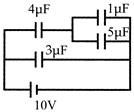
The charge on \[4\mu F\] capacitor in the given circuit is: \[(in\,\mu c)\]
A)
12
done
clear
B)
24
done
clear
C)
36
done
clear
D)
32
done
clear
View Answer play_arrow
Find the inductance L of a solenoid of length\[\ell \]whose winding are made of material of density D and resistivity\[\rho \]. The winding resistance is R:
A)
\[\frac{{{\mu }_{{}^\circ }}Rm}{4\pi R\rho D}\]
done
clear
B)
\[\frac{{{\mu }_{{}^\circ }}\ell m}{4\pi R\rho D}\]
done
clear
C)
\[\frac{{{\mu }_{{}^\circ }}}{4\pi \ell }\frac{{{R}^{2}}m}{\rho D}\]
done
clear
D)
\[\frac{{{\mu }_{{}^\circ }}}{2\pi \ell }\frac{\ell \,m}{\rho D}\]
done
clear
View Answer play_arrow
Two particles of masses \[{{m}_{1}}\] and \[{{m}_{2}}\] are connected to a rigid massless rod of length r to constitute a dumb bell which is free to move in the plane. The moment of inertia of the dumb bell about an axis perpendicular to the plane passing through the centre of mass is:
A)
\[\frac{{{m}_{1}}{{m}_{2}}}{{{m}_{1}}+{{m}_{2}}}{{r}^{2}}\]
done
clear
B)
\[({{m}^{1}}+{{m}^{2}}){{r}^{2}}\]
done
clear
C)
\[\frac{{{m}_{1}}{{m}_{2}}}{{{m}_{1}}-{{m}_{2}}}{{r}^{2}}\]
done
clear
D)
\[({{m}^{1}}-{{m}^{2}}){{r}^{2}}\]
done
clear
View Answer play_arrow
A molecule with dipole moment 'p' is placed in an electric field of strength E. Initially the dipole is aligned parallel to the field If the dipole is to be rotated to be antiparallel to the field, the work required to De done by an external agency is:
A)
? 2pE
done
clear
B)
? pE
done
clear
C)
pE
done
clear
D)
2Pe
done
clear
View Answer play_arrow
A prims having refractive index 1.414 and refracting angle \[30{}^\circ \]has one of the refracting surfaces silvered A beam of light incident on the other refracting surface will retrace its path, if the angle of incidence is:
A)
\[0{}^\circ \]
done
clear
B)
\[\frac{\pi }{6}\]
done
clear
C)
\[\frac{\pi }{3}\]
done
clear
D)
\[\frac{\pi }{4}\]
done
clear
View Answer play_arrow
A parallel beam of fast moving electrons is incident normally on a narrow slit. A fluorescent screen is placed at a large distance from the slit. If the speed of the electrons is increased, then which of the following is correct?
A)
Diffraction pattern is not observed on the screen in the case of electrons
done
clear
B)
The angular width of the central maximum of the diffraction pattern will increase
done
clear
C)
The angular width of the central maximum will decrease
done
clear
D)
The angular width of the central maxi- mum will be unaffected
done
clear
View Answer play_arrow
A rectangular coil of 20 turns and cross section area \[25c{{m}^{2}}\] has a resistance of 100Q. If a magnetic field which is perpendicular to the plane of the coil changes at a rate of 1000T/s, the current in the coil is:
A)
1A
done
clear
B)
50A
done
clear
C)
0.5A
done
clear
D)
5A
done
clear
View Answer play_arrow
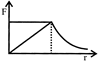
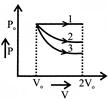
A gas is expanded from volume \[{{V}_{{}^\circ }}\] to \[2{{V}_{{}^\circ }}\] under three different processes, as shown in the figure. Process 1 is isobaric process, process 2 is isothermal and process 3 is adiabatic Let \[\Delta \,{{U}_{1}}\], \[\Delta \,{{U}_{2}}\] and \[\Delta \,{{U}_{3}}\] be the change in the internal energy of the gas in these three processes, then:
A)
\[\Delta {{U}_{1}}>\Delta {{U}_{2}}>\Delta {{U}_{3}}\]
done
clear
B)
\[\Delta {{U}_{2}}<\Delta {{U}_{1}}<\Delta {{U}_{3}}\]
done
clear
C)
\[\Delta {{U}_{1}}<\Delta {{U}_{2}}<\Delta {{U}_{3}}\]
done
clear
D)
\[\Delta {{U}_{2}}<\Delta {{U}_{3}}<\Delta {{U}_{1}}\]
done
clear
View Answer play_arrow
Same force acts on two bodies of different masses 3kg and 5kg initially at rest. The ratio of time required to acquire same final velocity is:
A)
5 : 3
done
clear
B)
25 : 9
done
clear
C)
9 : 25
done
clear
D)
3 : 5
done
clear
View Answer play_arrow
A ball whose density is \[0.4\times 10g/{{m}^{3}}\]falls into water from a height of 9cm. To what depth does the ball sink?
A)
9cm
done
clear
B)
6cm
done
clear
C)
4.5cm
done
clear
D)
2.25cm
done
clear
View Answer play_arrow
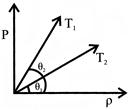
The figure shows pressure versus density graph for an ideal gas at two temperature \[{{T}_{1}}\] and \[{{T}_{2}}\]:
A)
\[{{T}_{1}}>{{T}_{2}}\]
done
clear
B)
\[{{T}_{1}}={{T}_{2}}\]
done
clear
C)
\[{{T}_{1}}<{{T}_{2}}\]
done
clear
D)
\[None\]
done
clear
View Answer play_arrow
A string of length L and mass M is lying on a horizontal table. A force F is applied at one of its ends. Tension in the string at a distance y from the end at which the force applied is:
A)
\[Zero\]
done
clear
B)
\[F\]
done
clear
C)
\[\frac{F\,(L-Y)}{L}\]
done
clear
D)
\[\frac{F\,(L-Y)}{M}\]
done
clear
View Answer play_arrow
The prefix \[{{10}^{18}}\] is:
A)
Giga
done
clear
B)
Exa
done
clear
C)
Kilo
done
clear
D)
Nano
done
clear
View Answer play_arrow
Identify the least stable among the following:
A)
\[L{{i}^{-}}\]
done
clear
B)
\[B{{e}^{-}}\]
done
clear
C)
\[{{B}^{-}}\]
done
clear
D)
\[{{C}^{-}}\]
done
clear
View Answer play_arrow
EKa- aluminium and EKa-silicon are known as:
A)
Gallium and germanium
done
clear
B)
Aluminum and silicon
done
clear
C)
Iron and sulphur
done
clear
D)
Proton and silicon
done
clear
View Answer play_arrow
Which of the following transitions will have minimum wavelength?
A)
\[{{n}_{4}}\to {{n}_{1}}\]
done
clear
B)
\[{{n}_{2}}\to {{n}_{1}}\]
done
clear
C)
\[{{n}_{4}}\to {{n}_{2}}\]
done
clear
D)
\[{{n}_{3}}\to {{n}_{1}}\]
done
clear
View Answer play_arrow
The molecule/species having one unpaired electron is:
A)
\[NO\]
done
clear
B)
\[CO\]
done
clear
C)
\[C{{N}^{-}}\]
done
clear
D)
\[{{O}_{2}}\]
done
clear
View Answer play_arrow
Which one of the following has highest dipole moment?
A)
\[N{{H}_{3}}\]
done
clear
B)
\[P{{H}_{3}}\]
done
clear
C)
\[Sb{{H}_{3}}\]
done
clear
D)
\[As{{H}_{3}}\]
done
clear
View Answer play_arrow
Hydrogen bonding is maximum in:
A)
Ethanol
done
clear
B)
Diethyl ether
done
clear
C)
Ethyl chloride
done
clear
D)
Triethlamine
done
clear
View Answer play_arrow
In \[Br{{F}_{3}}\] molecule the lone pairs occupy equatorial position to minimize:
A)
Lone pair - bond pair repulsion only
done
clear
B)
Bond pair - bond pair repulsion only
done
clear
C)
Lone pair - lone pair repulsion and lone pair bond pair repulsion
done
clear
D)
Lone pair - lone pair repulsion only
done
clear
View Answer play_arrow
The following graph illustrates:
A)
Dalton's law
done
clear
B)
Charle's law
done
clear
C)
Boyle's law
done
clear
D)
Gay lussac's law
done
clear
View Answer play_arrow
How much energy is released when 6 moles of octane is burnt in air? Given \[\Delta H_{f}^{{}^\circ }\] for \[C{{O}_{2}}(g)\,,{{H}_{2}}O(g)\]and respectively are - 490, - 240 and+160kJ/mol:
A)
- 6.2kJ
done
clear
B)
- 37.4kJ
done
clear
C)
- 35.5kJ
done
clear
D)
- 20.0kJ
done
clear
View Answer play_arrow
9.2 grams of is taken in a closed one litre vessel and heated fill the following equilibrium is reached\[{{N}_{2}}{{O}_{4}}(g)\,\,2N{{O}_{2}}(g)\,\]. At equilibrium is dissociated what is the equilibrium constant (in mol/ L) \[(molecular\,\,weight\,\,of\,{{N}_{2}}{{O}_{4}}=92)\]
A)
0.1
done
clear
B)
0.2
done
clear
C)
0.4
done
clear
D)
2
done
clear
View Answer play_arrow
The pKa of a weak acid is 4.8. What should be the ratio of [Acid] / [Salt] of a buffer is pH = 5.8 is repaired?
A)
0.1
done
clear
B)
10
done
clear
C)
1
done
clear
D)
2
done
clear
View Answer play_arrow
\[{{E}^{{}^\circ }}F{{e}^{2+}}/=\,Fe=-\,0.441V\] and \[{{E}^{{}^\circ }}{{F}^{3+}}=0.771V\] the standard EMF of the reaction, \[Fe+2F{{e}^{3+}}\to 3{{F}^{2+}}\]will be:
A)
0.111V
done
clear
B)
0.330V
done
clear
C)
1.653V
done
clear
D)
1.212V
done
clear
View Answer play_arrow
Para and or the hydrogen differ in:
A)
Atomic number
done
clear
B)
Atomic mass
done
clear
C)
Spins of protons
done
clear
D)
Number of neutron
done
clear
View Answer play_arrow
Solubilities of carbonates decrease down the magnesium group due to decease in:
A)
Entropy of solution formation
done
clear
B)
Lattice energies of solids
done
clear
C)
Hydration energy of cations
done
clear
D)
Inter-ionic attraction
done
clear
View Answer play_arrow
The charge/size ratio of a action determines its polarizing power. Which one of the following sequences represents the increasing order of polarizing power of cationic species, \[{{K}^{+}},\,C{{a}^{2+}},M{{g}^{2+}},B{{e}^{2+}}\]
A)
\[C{{a}^{2+}}<M{{g}^{2+}}<B{{e}^{2+}}<{{K}^{+}}\]
done
clear
B)
\[M{{g}^{2+}}<\,\,B{{e}^{2+}}<{{K}^{+}}<C{{a}^{2+}}\]
done
clear
C)
\[B{{e}^{2+}}<{{K}^{+}}<C{{a}^{2+}}<M{{g}^{2+}}\]
done
clear
D)
\[{{K}^{+}}<C{{a}^{2+}}<M{{g}^{2+}}<B{{e}^{2+}}\]
done
clear
View Answer play_arrow
The C-H bond distance is longest in:
A)
\[{{C}_{2}}{{H}_{2}}\]
done
clear
B)
\[{{C}_{2}}{{H}_{4}}\]
done
clear
C)
\[{{C}_{2}}{{H}_{6}}\]
done
clear
D)
\[{{C}_{2}}{{H}_{2}}B{{r}_{2}}\]
done
clear
View Answer play_arrow
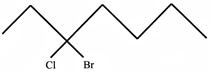
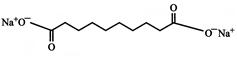
The IUPAC name of the following compound is:
A)
1-Bromo-3-Chlorohexane
done
clear
B)
2-Bromo-3-Chloroheptane
done
clear
C)
3-Bromo, Chloroheptane
done
clear
D)
3-Bromo-3-Chloroheptane
done
clear
View Answer play_arrow
Which of the following represents the given mode of hybridisation \[s{{p}^{2}}-s{{p}^{2}}-sp-sp\] from left to right?
A)
\[{{H}_{2}}C=CH-C\equiv Na\]
done
clear
B)
\[HC\equiv C-C\equiv CH\]
done
clear
C)
\[{{H}_{2}}C=C=C=C{{H}_{2}}\]
done
clear
D)
done
clear
View Answer play_arrow
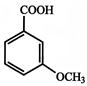
Which of the following compounds is not aromatic?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
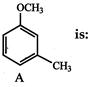
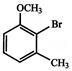
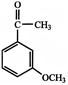
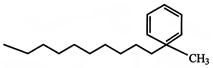
The major product obtained on monobromination \[(B{{r}_{2}}/FeB{{r}_{3}})\] of the compound A is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
\[BO{{D}_{5}}\]is:
A)
Waste decomposed in 5 days
done
clear
B)
Oxygen used in 5 days
done
clear
C)
Microorganisms killed in 5 days
done
clear
D)
Dissolved oxygen left after 5 days
done
clear
View Answer play_arrow
In a tetragonal crystal:
A)
\[\alpha =b=c,\alpha =\beta =90{}^\circ =\gamma \]
done
clear
B)
\[\alpha =\beta =\gamma =90{}^\circ ,\text{ }\alpha =b\ne c\]
done
clear
C)
\[\alpha =\beta =\gamma =90{}^\circ ,\text{ }a\ne b\ne c\]
done
clear
D)
\[\alpha =\beta =90{}^\circ ,\text{ }\gamma =120{}^\circ ,\text{ }a=b\ne c\]
done
clear
View Answer play_arrow
The pyknometric density of sodium chloride crystal is \[2.165\times {{10}^{3}}kg{{m}^{-3}}\] while its x-ray density is\[2.178\times {{10}^{3}}\,Kg{{m}^{-3}}\]. The fraction of unoccupied sites in sodium chloride crystal is:
A)
\[5.96\]
done
clear
B)
\[5.96\times {{10}^{-2}}\]
done
clear
C)
\[5.96\times {{10}^{-1}}\]
done
clear
D)
\[5.96\times {{10}^{-3}}\]
done
clear
View Answer play_arrow
An X molal solution of a compound in benzene has mole fraction of solute equal to 0.2. The value of X is nearly:
A)
14
done
clear
B)
3.2
done
clear
C)
1.4
done
clear
D)
2
done
clear
View Answer play_arrow
An aqueous solution freezes at \[-\,0.186{}^\circ \,C\,({{K}_{f}}=1.86{}^\circ ;{{K}_{b}}=0.512{}^\circ ).\]What is the elevation in boiling point?
A)
0.186
done
clear
B)
0.512
done
clear
C)
0.86
done
clear
D)
0.0512
done
clear
View Answer play_arrow
The standard emf of the \[Cd\,(s)/Cd\text{ }Cl,(0.1)\,M//\text{ }Ag\,Cl\,(s)/Ag\,(s)\] in which cell reaction is \[+\,\,2C{{l}^{-}}\,(aq.)\text{ }\]is \[0.6915v\text{ }at\text{ }0{}^\circ C\] and\[0.6753v\text{ }at25{}^\circ .C\]. The enthalpy change of the reaction at\[25{}^\circ C\]is:
A)
(a)\[-176kJ\]
done
clear
B)
\[-\,334.7kJ\]
done
clear
C)
\[+123.5kJ\]
done
clear
D)
\[-\,167.6kJ\]
done
clear
View Answer play_arrow
For an endothermic reaction, where \[\Delta H\] represents the enthalpy of the reaction in kJ/mol, the minimum value for the energy of activation will be:
A)
Less than \[\Delta H\]
done
clear
B)
Zero
done
clear
C)
More than \[\Delta H\]
done
clear
D)
Equal to \[\Delta H\]
done
clear
View Answer play_arrow
In reversible reaction, \[2\,N{{O}_{2}}{{N}_{2}}{{O}_{4}}\] the rate of disappearance on \[N{{o}_{2}}\] is equal to:
A)
\[\frac{2{{K}_{1}}}{{{K}_{2}}}\,{{[N{{O}_{2}}]}^{2}}\]
done
clear
B)
\[2\,{{K}_{1}}{{[N{{O}_{2}}]}^{2}}-\,2{{K}_{2}}[{{N}_{2}}{{O}_{4}}]\]
done
clear
C)
\[2\,{{K}_{1}}{{[N{{O}_{2}}]}^{2}}-\,{{K}_{2}}[{{N}_{2}}{{O}_{4}}]\]
done
clear
D)
\[(2{{K}_{1}}-\,\,{{K}_{2}})\,[N{{O}_{2}}]\]
done
clear
View Answer play_arrow
Which of the following forms cationic micelles above certain concentration?
A)
Sodium dodecyl sulphates
done
clear
B)
Sodium acetate
done
clear
C)
Urea
done
clear
D)
Cetyl trim ethyl ammonium bromide
done
clear
View Answer play_arrow
Vapour phase refining of nickel is carried out using:
A)
\[{{I}_{2}}\]
done
clear
B)
\[C{{I}_{2}}\]
done
clear
C)
\[HCI\]
done
clear
D)
\[CO\]
done
clear
View Answer play_arrow
The number of hydrogen atoms attached to phosphorus atom in hypo phosphorus acid in:
A)
Zero
done
clear
B)
Two
done
clear
C)
One
done
clear
D)
Three
done
clear
View Answer play_arrow
The number of P-O-P bonds in the structure of phosphorus pentoxide and phosphorus trioxide are respectively:
A)
6, 6
done
clear
B)
5, 5
done
clear
C)
5, 6
done
clear
D)
6, 5
done
clear
View Answer play_arrow
Number of moles of \[{{K}_{2}}C{{r}_{2}}{{O}_{7}}\], reduced by one mole of \[S{{n}^{2+}}\] ions is:
A)
1/3
done
clear
B)
3
done
clear
C)
1/6
done
clear
D)
6
done
clear
View Answer play_arrow
The compound used for gravimetric estimation of Cu(II) is:
A)
\[C{{u}_{2}}{{(SCN)}_{2}}\]
done
clear
B)
\[C{{u}_{2}}O\]
done
clear
C)
\[C{{u}_{2}}{{I}_{2}}\]
done
clear
D)
\[C{{u}_{2}}C{{O}_{3}}\]
done
clear
View Answer play_arrow
The pair in which both species have same magnetic moment (spin only value) is:
A)
\[{{[Cr{{({{H}_{2}}O)}_{6}}]}^{2+}},\,\,{{[COC{{l}_{4}}]}^{2-}}\]
done
clear
B)
\[{{[Cr{{({{H}_{2}}O)}_{6}}]}^{2+}},\,\,{{[Fe{{({{H}_{2}}O)}_{6}}]}^{2+}}\]
done
clear
C)
\[{{[Mn{{({{H}_{2}}O)}_{6}}]}^{2+}},\,\,{{[Cr{{({{H}_{2}}O)}_{6}}]}^{2+}}\]
done
clear
D)
\[{{[COC{{l}_{4}}]}^{2-}},\,\,{{[Fe{{({{H}_{2}}O)}_{6}}]}^{2+}}\]
done
clear
View Answer play_arrow
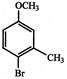
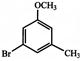
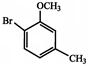
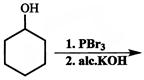
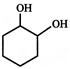
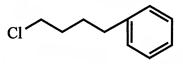
Which of the following sequence of reactions (reagents) can be used for conversion of
A)
\[SOC{{l}_{2}};\,{{H}_{2}}O\]
done
clear
B)
\[S{{O}_{2}}C{{l}_{2}};\,alc\,KOH\]
done
clear
C)
\[C{{l}_{2}}/;hv\,{{H}_{2}}O\]
done
clear
D)
\[SOC{{l}_{2}};\,alc.KOH\]
done
clear
View Answer play_arrow
Predict the product:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A strong base can abstract an \[\alpha \]-hydrogen from:
A)
Ketone
done
clear
B)
Alkane
done
clear
C)
Alkene
done
clear
D)
Amine
done
clear
View Answer play_arrow
The product p in the reaction:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
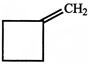
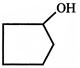
Treatment of cyclobutyl methyamine with nitrous acid does not give:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
In \[H{{S}^{-}},{{I}^{-}},R-N{{H}_{2}},N{{H}_{3}},\] the order of proton accepting tendency will be:
A)
\[{{I}^{-}}>N{{H}_{3}}>R-N{{H}_{2}}>H{{S}^{-}}\]
done
clear
B)
\[N{{H}_{3}}>R-N{{H}_{2}}>H{{S}^{-}}>{{I}^{-}}\]
done
clear
C)
\[R-N{{H}_{2}}>N{{H}_{3}}>H{{S}^{-}}>{{I}^{-}}\]
done
clear
D)
\[H{{S}^{-}}>R-N{{H}_{2}}>N{{H}_{3}}>{{I}^{-}}\]
done
clear
View Answer play_arrow
The secondary structure of a protein refers to:
A)
Fixed configuration of polypeptide back bone
done
clear
B)
a- helical backbone
done
clear
C)
Hydrophobic interactions
done
clear
D)
Sequence of a-amino acids
done
clear
View Answer play_arrow
Which of the following is polyamide?
A)
Teflon
done
clear
B)
Nylon-6, 6
done
clear
C)
Terylene
done
clear
D)
Bakelite
done
clear
View Answer play_arrow
Which of the following can disperse in water:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following statement is correct?
A)
In plants, growth by cell division occurs continuously throughout their life span
done
clear
B)
Non-living objects grow by accumulation of material on the surface
done
clear
C)
In unicellular organisms, growth & repro-duction are same
done
clear
D)
Isolated metabolic reactions in vitro are living things but are not living reactions.
done
clear
View Answer play_arrow
Which of the following is incorrect matching:
A)
Rhizopus - Bread mould
done
clear
B)
Albugo - Parasitic fungi on mustard
done
clear
C)
Colletotrichun - Imperfect fungi
done
clear
D)
Puff balls - Ascocarps
done
clear
View Answer play_arrow
A great role in recycling nutrients like nitrogen, phosphorus, iron and sulphur:
A)
Chemosynthetic autotrophs
done
clear
B)
Heterotrophic bacteria
done
clear
C)
Photosynthetic autotrophs
done
clear
D)
Cyanobacteria
done
clear
View Answer play_arrow
The chief 'producers' in the ocean are:
A)
Dinoflagellates
done
clear
B)
Diatoms
done
clear
C)
Euglena
done
clear
D)
Ganyaulax
done
clear
View Answer play_arrow
Which of the following is incorrect statement?
A)
Mesosome is formed by the infolding of plasma membrane
done
clear
B)
All cells contain non-membrane bound organelles called Ribosome
done
clear
C)
Bacterial flagellum is made of 2 parts
done
clear
D)
Chromatophores are membraneous in-folding in some prokaryotes
done
clear
View Answer play_arrow
In human RBC, membrane has ____protein & ____ lipids:
A)
50% protein & 50% lipids
done
clear
B)
52% protein & 46% lipids
done
clear
C)
42% protein & 48% lipids
done
clear
D)
52% protein & 40% lipids
done
clear
View Answer play_arrow
Term Omnis cellula - e - cellula was given by:
A)
Rudolf virchow (1865)
done
clear
B)
Rudolf virchow (1895)
done
clear
C)
Rudolf virchow (1875)
done
clear
D)
Rudolf virchow (1855)
done
clear
View Answer play_arrow
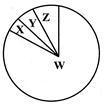
Identify W, X, Y and Z w.r.t biodiversity among invertebrates:
A)
W
X
Y
Z
Insects
Molluscs
Crustaceans
Other mammals
done
clear
B)
W
X
Y
Z
Molluscs
Insects
Other inver- tebrates
Crusta- ceans
done
clear
C)
W
X
Y
Z
Insects
Molluscs
Crustaceans
Other inverte- brates
done
clear
D)
W
X
Y
Z
Insects
Other inverte- brates
Crustaceans
Molluscs
done
clear
View Answer play_arrow
Protein synthesized by ribosomes on the ER are modified in the:
A)
Stroma of the Golgi apparatus before they are released from its trans face.
done
clear
B)
Cisternae of the Golgi apparatus before they are released from its trans face
done
clear
C)
Lumen of the Golgi apparatus before they are released from its trans face
done
clear
D)
Cisternae of the Golgi apparatus after they are released from its trans face
done
clear
View Answer play_arrow
Fusion between one large, non-motile (static) female gamete and a smaller, motile mate gamete seen in:
A)
Fucus
done
clear
B)
Volvox
done
clear
C)
Both
done
clear
D)
Chlamydomonas
done
clear
View Answer play_arrow
Which of the following is a set of hydrocolloids?
A)
Algin (Red algae) & Carragean (Brown algae)
done
clear
B)
Algin (Brown algae) & Carragean (Red algae)
done
clear
C)
Algin (Brown algae) only
done
clear
D)
Carrageen (Brown algae) only
done
clear
View Answer play_arrow
Which of the following is not comes under Numerical taxonomy:
A)
Based on all observable characteristics
done
clear
B)
Number & codes are assigned to all the characters and the data are processed
done
clear
C)
Uses the chemical constituents of the plant
done
clear
D)
Hundreds of characters can be considered giving equal importance
done
clear
View Answer play_arrow
Fleshy leaves are present in:
A)
Alstonia
done
clear
B)
Garlic
done
clear
C)
Cacti
done
clear
D)
Peas
done
clear
View Answer play_arrow
The cells differentiate and mature in:
A)
Region of maturation
done
clear
B)
Region of meristematic activity
done
clear
C)
Region of elongation
done
clear
D)
oot cap
done
clear
View Answer play_arrow
A green flattened or fleshy cylindrical stem containing chlorphyll for photosynthesis:
A)
Phyllociade
done
clear
B)
Thorns
done
clear
C)
Stem tendrils
done
clear
D)
Phyllode
done
clear
View Answer play_arrow
Monoadelphous condition is found in:
A)
Pea
done
clear
B)
China rose
done
clear
C)
Citrus
done
clear
D)
Mustard
done
clear
View Answer play_arrow
Reduced of leaf found at the base of the pedice is called:
A)
Pedicel
done
clear
B)
Receptacle
done
clear
C)
Thalamus
done
clear
D)
Bracts
done
clear
View Answer play_arrow
The government of India has passed the environment (protection) act on?
A)
1987
done
clear
B)
1986
done
clear
C)
1985
done
clear
D)
1984
done
clear
View Answer play_arrow
Which of the following is incorrect statement?
A)
Particulates size of less than PM 2.5 causes greatest harm to human health
done
clear
B)
Air pollutant cause injury to all living organisms
done
clear
C)
A scrubber removes gases like \[S{{O}_{2}}\]
done
clear
D)
Acute exposure to relatively lower noise may damage hearing abilities of humans
done
clear
View Answer play_arrow
Poly blend is mixed with:
A)
Butene
done
clear
B)
Butane
done
clear
C)
Methane
done
clear
D)
Bituman
done
clear
View Answer play_arrow
The living state is a:
A)
Equilibrium steady - state to be able to perform work
done
clear
B)
Non-equilibrium steady - state to be able to perform work
done
clear
C)
May be both time to time
done
clear
D)
Thermodynamically steady state
done
clear
View Answer play_arrow
India initiated reproductive health programs (family planning) in:
A)
1947
done
clear
B)
1984
done
clear
C)
1985
done
clear
D)
1951
done
clear
View Answer play_arrow
In periodic abstinence:
A)
Avoid coitus from day 10 to 17 of the menstrual cycle (fertile period) ?
done
clear
B)
Do coitus daily
done
clear
C)
No chances of formation of zygote
done
clear
D)
Withdraw penis from the vagina just before ejaculation
done
clear
View Answer play_arrow
'Bad' ozone is formed in:
A)
Lower atmosphere (troposphere)
done
clear
B)
Upper atmosphere (troposphere)
done
clear
C)
Lower atmosphere (stratosphere)
done
clear
D)
Upper atmosphere (stratosphere)
done
clear
View Answer play_arrow
Choose the incorrect one:
A)
Cyclosporine A: Trichoderma polysporum
done
clear
B)
Statins: Monascus purpureus
done
clear
C)
Streptokinase: Strepto coccus
done
clear
D)
Aspergillus niger: Acetic acid
done
clear
View Answer play_arrow
Baculoviruses attacks on:
A)
Arthropods only
done
clear
B)
Insects & other arthropods
done
clear
C)
Insects only
done
clear
D)
Bacteriophages
done
clear
View Answer play_arrow
Duration of yeast cell cycle is about:
A)
50 Minutes
done
clear
B)
1 hour
done
clear
C)
120 Minutes
done
clear
D)
90 minutes
done
clear
View Answer play_arrow
X-shaped structures are called chiasmata formed in which stage:
A)
Diakinesis
done
clear
B)
Diplotene
done
clear
C)
Pachytene
done
clear
D)
Leptotene
done
clear
View Answer play_arrow
Thyrocalcitonin regulates:
A)
Blood calcium level
done
clear
B)
Metabolism of carbohydrates
done
clear
C)
Basal metabolic rate (BMR)
done
clear
D)
Maintenance of water and electrolyte balance
done
clear
View Answer play_arrow
Regulate growth of mammary glands:
A)
Oxytocin
done
clear
B)
Prolactin
done
clear
C)
Growth hormone
done
clear
D)
LH
done
clear
View Answer play_arrow
Which one is not the function of Glucocorticoid:
A)
Maintain Cardio vascular system and kidney functions
done
clear
B)
Cortisol stimulates RBC production
done
clear
C)
Produces anti-inflammatory reactions and suppress immune response
done
clear
D)
Growth of axial hair, public hair and facial hair during puberty
done
clear
View Answer play_arrow
I-band is bisected by a dense dark band called:
A)
Z - line
done
clear
B)
M - line
done
clear
C)
H - line
done
clear
D)
K - line
done
clear
View Answer play_arrow
Select incorrect statement:
A)
Number of cervical vertebrate are in almost all mammals
done
clear
B)
Vertebrate has a central hollow portion (neural canal) through which the spinal cord passes
done
clear
C)
Each coxal bone is formed by the fusion of two bones
done
clear
D)
Joint between carpel & metacarpal of thumb called saddle joints
done
clear
View Answer play_arrow
Choose incorrect matching:
A)
Gemmule - sponge
done
clear
B)
Conidia - penicillium
done
clear
C)
Adventitious buds - Bryophyllum
done
clear
D)
Multiple fission - Planaria
done
clear
View Answer play_arrow
Occur in grasses and regenerate parts removed by the grazing herbivore:
A)
Axillary bud
done
clear
B)
Primary meristems
done
clear
C)
Intercalary meristems
done
clear
D)
Sclereids
done
clear
View Answer play_arrow
Pit is large & well developed in:
A)
Dicot root
done
clear
B)
Monocot root
done
clear
C)
Monocot shoot
done
clear
D)
Dicot shoot
done
clear
View Answer play_arrow
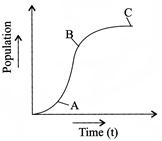
What do A, B and C represent in the diagram:
A)
Stationary, log and lag
done
clear
B)
Lag, stationary and log
done
clear
C)
Log, lag and stationary
done
clear
D)
Lag, log and stationary
done
clear
View Answer play_arrow
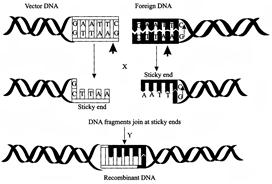
Identify the enzyme X and Y?
A)
X=Sma I, Y=DNA ligase
done
clear
B)
X = Barn HI, Y = RNA-ligase
done
clear
C)
X = Eco RI, Y = DNA-ligase
done
clear
D)
X = Hind III, Y = RNA-ligase
done
clear
View Answer play_arrow
Protect against chemical & mechanical stresses:
A)
Compound epithelium
done
clear
B)
Ciliated epithelium
done
clear
C)
Cuboidal epithelium
done
clear
D)
Columnar epithelium
done
clear
View Answer play_arrow
Which of the following is not a feature of male cockroach?
A)
Anal styles present
done
clear
B)
Broad pouch absent
done
clear
C)
Broad abdonors
done
clear
D)
Large in size
done
clear
View Answer play_arrow
Select the incorrect statement:
A)
About 3% of \[{{O}_{2}}\] is carried in a dissolved state through plasma
done
clear
B)
Every 100ml of deoxygenated blood delivers about 4ml of \[C{{O}_{2}}\] to the alveoli
done
clear
C)
In emphysema alveolar walls are damaged
done
clear
D)
In tissues, low\[P{{O}_{2}}\], high p\[C{{O}_{2}}\], low H+ ions and High temperature exist. So\[H{{b}_{4}}{{O}_{8}}\] dissociates to release \[{{O}_{2}}\]
done
clear
View Answer play_arrow
Which of the following is a manmade ecosystem?
A)
Pond
done
clear
B)
Lake
done
clear
C)
Wetland
done
clear
D)
Aquarium
done
clear
View Answer play_arrow
Decomposition rate is slower if detritus is rich in:
A)
Lignin & Chitin
done
clear
B)
Nitrogen & Sugars
done
clear
C)
Nitrogen & Chitin
done
clear
D)
Sugars & Lignin
done
clear
View Answer play_arrow
Sun is the only source of energy for all ecosystem except:
A)
Desert
done
clear
B)
Amazone forest
done
clear
C)
Tropical rain forests
done
clear
D)
Deep sea hydro - thermal ecosystem
done
clear
View Answer play_arrow
Select incorrect one:
A)
Substances soluble in lipids diffuse through the membrane faster
done
clear
B)
Pumps are proteins that use energy to transport substance across cell membrane
done
clear
C)
Water is a universal solvent essential for all physiological activities of living organisms
done
clear
D)
Water molecules move from lower water potential to higher water potential
done
clear
View Answer play_arrow
The suberin in the root endodermis allows the ____ transport of ions direction only:
A)
Passive
done
clear
B)
Active
done
clear
C)
Facillitated
done
clear
D)
Diffusion
done
clear
View Answer play_arrow
Select incorrect one:
A)
Mucous neck cells: secrete mucous
done
clear
B)
Chief cells: secrete inactive enzymes like pepsinogen, prorinnin and lipase
done
clear
C)
Zymogen or peptic cells: Secrete Hcl
done
clear
D)
Oxyntic (parietal) cells: Secrete Hcl & castle's gastric
done
clear
View Answer play_arrow
Which of the following is not present in bile juice?
A)
Biliverdin
done
clear
B)
Phospholipids
done
clear
C)
Chlolesterol
done
clear
D)
Enzymes
done
clear
View Answer play_arrow
Sperms stores temporarily in:
A)
Rete testis
done
clear
B)
Seminal vesicle
done
clear
C)
Epididymis
done
clear
D)
Urethra
done
clear
View Answer play_arrow
Choose the wrong statement:
A)
The lumen of vegina is lined by a glycogen rich mucous membrane consisting of sensitive papillae and Bartholdi?s gland
done
clear
B)
Mons pubis is a cushion of fatty tissue covered by pubic hair
done
clear
C)
Clitoris is a highly sensitive organ lying just in front of the urethral opening
done
clear
D)
Maiden head also known as prepuce
done
clear
View Answer play_arrow
Innermost layer of ovum is known as:
A)
Oolemma
done
clear
B)
Vitelline membrane
done
clear
C)
Zona pellucida
done
clear
D)
Corona radiata
done
clear
View Answer play_arrow
It is used to speed up malting process in brewing industary:
A)
Auxin
done
clear
B)
ABA
done
clear
C)
Gibberellin
done
clear
D)
Ethylene
done
clear
View Answer play_arrow
Heterophylly due to environment is present in:
A)
Cotton
done
clear
B)
Coriander
done
clear
C)
Larkspur
done
clear
D)
Ranunculus
done
clear
View Answer play_arrow
Select the wrong statement:
A)
Plasma without clothing factors is known as serum
done
clear
B)
If bloods with interactive antigens and antibodies are mixed together, it causes clumping (agglutination) of RBCs
done
clear
C)
Largest WBC are Neutrophils
done
clear
D)
Incomplete double circulation present in amphibians & reptiles
done
clear
View Answer play_arrow
Scientist who described pumping action of heart and blood pathway in venis and arteries is:
A)
Ernst Mayr
done
clear
B)
Alexender fleming
done
clear
C)
William Harvey
done
clear
D)
James Watson
done
clear
View Answer play_arrow
Theory of Biogenesis was proposed by:
A)
Francisco Redi, spallanzani & Louis pos-teur
done
clear
B)
Oparin & Haldane
done
clear
C)
Harold urey & Stanley miller
done
clear
D)
Y. D. sharma
done
clear
View Answer play_arrow
The process by which unrelated species become more similar in order to survive and adapt in similar environmental conditions:
A)
Homology
done
clear
B)
Analogy
done
clear
C)
Both
done
clear
D)
Adaptive radiation
done
clear
View Answer play_arrow
Choose the correct combination:
A)
Uricotelism: Cartilaginous fishes
done
clear
B)
Ammonotelism: Land snails
done
clear
C)
Ureotelism: Tadpoles
done
clear
D)
Green gland: Crustaceans
done
clear
View Answer play_arrow
Vasa recta are absent or highly reduced in:
A)
Cortical nephrons
done
clear
B)
Juxta medullary nephrons
done
clear
C)
In both
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which of the following is a set of Fresh water fishes?
A)
Hilsa & Sardines
done
clear
B)
Mackerel & Catia
done
clear
C)
Rohu & Common carp
done
clear
D)
Rohu & Sardines
done
clear
View Answer play_arrow
Taichung Native -1 is a:
A)
Semi - dwarf wheat varieties
done
clear
B)
Semi - dwarf rice varieties
done
clear
C)
Semi - dwarf rye varieties
done
clear
D)
Semi - dwarf maize varieties
done
clear
View Answer play_arrow
Glucose & fructose are phosphorylated to form & glucose 6 - phosphate by the enyme:
A)
Helicase
done
clear
B)
Hexokinase
done
clear
C)
Invertase
done
clear
D)
Glucase
done
clear
View Answer play_arrow
GTP molecule is synthesized when:
A)
Succinyl - CoA is converted to succinic acid
done
clear
B)
Citrate is isomerizes to Isocitrate
done
clear
C)
Condensation of acetyl group with OAA
done
clear
D)
All of these
done
clear
View Answer play_arrow
A)
(a) Anthropoda Joint - legged animals Locusta
done
clear
B)
(b) Echinodermata Spiny skined animal Ophiura
done
clear
C)
(c) Annelida Segmented worms Pheretima
done
clear
D)
(d) Aschelmithes Sexual dimorphism Balaenoptera
done
clear
View Answer play_arrow
Pentadactyl limbs is a feature of:
A)
Chondricthyes
done
clear
B)
Osteichthyes
done
clear
C)
Amphibia
done
clear
D)
Reptila
done
clear
View Answer play_arrow
Skin is dry without glands except the oil gland at the base of the tail in:
A)
Aptenodytes
done
clear
B)
Pterophyllum
done
clear
C)
Equus
done
clear
D)
Trygon
done
clear
View Answer play_arrow
Retrogressive metamorphosis is a feature of:
A)
Cephalochordata
done
clear
B)
Craniata
done
clear
C)
Tunicata
done
clear
D)
Vertebrata
done
clear
View Answer play_arrow
Pick out wrong statement:
A)
Out of every 10 animals, tare insects
done
clear
B)
India has only 2.4% of world's land area
done
clear
C)
Species diversity increases from the equator to the poles
done
clear
D)
Biodiversity is highest in tropics
done
clear
View Answer play_arrow
For frugivorous birds and mammals in the tropical forests of different continent, the slope is:
A)
6.5
done
clear
B)
1.0
done
clear
C)
1.15
done
clear
D)
2.0
done
clear
View Answer play_arrow
Total number of Hotspot in the world is:
A)
34
done
clear
B)
20
done
clear
C)
30
done
clear
D)
15
done
clear
View Answer play_arrow
Identify A, B and C in the diagram depicting transcription below:
A)
a. Rho (p) protein RNA Polymerases Initiation
done
clear
B)
b. RNA Polymerases Initiation Rho (p) protein
done
clear
C)
c. Initiation RNA Polymerases Rho (p) protein
done
clear
D)
d. Rho(p) protein Initiation RNA Polymerases
done
clear
View Answer play_arrow
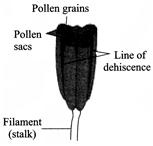
The diagram given below depicts an anther with:
A)
Monothecous + bisporangiate condition
done
clear
B)
Dithecous + tetrasporangiate condition
done
clear
C)
Dithecous + bisporangiate condition
done
clear
D)
Monothecous + tetrasporangiate condition
done
clear
View Answer play_arrow
Multipolar type of neurons is found in:
A)
Embryo
done
clear
B)
Cerebral cortex
done
clear
C)
Retina
done
clear
D)
Heart
done
clear
View Answer play_arrow
Choose the incorrect statement:
A)
Synapse is a functional between two neurons
done
clear
B)
Cerebellum also known as "little cerebrum".
done
clear
C)
The grey coloured area with myclin sheath is called grey matter
done
clear
D)
Hypothalamus controls food intake & blood pressure
done
clear
View Answer play_arrow
Organism tolerate a wide range of salinity:
A)
Euryhaline
done
clear
B)
Eurythermal
done
clear
C)
Stenothermal
done
clear
D)
Stenohaline
done
clear
View Answer play_arrow
99% of animals and nearly all plants cannot maintain a constant internal environment called:
A)
Regulators
done
clear
B)
Hibernation
done
clear
C)
Conformers
done
clear
D)
Diapauses
done
clear
View Answer play_arrow
Select the wrong statement:
A)
About 25% of all insects are phytophagous
done
clear
B)
Predators keep prey population under control
done
clear
C)
Biological control methods are based on the ability of predators to regulate prey population
done
clear
D)
Spines are the most common morphological means of defense of plants
done
clear
View Answer play_arrow
Dikaryophase means:
A)
2n per cell
done
clear
B)
n+n per cell
done
clear
C)
n+2n per cell
done
clear
D)
n per cell
done
clear
View Answer play_arrow
False feet is a feature of:
A)
Flagellated protozoans
done
clear
B)
Ciliated protozoans
done
clear
C)
Sporozoans
done
clear
D)
Amoeboid protozoans
done
clear
View Answer play_arrow
Select the incorrect one:
A)
In plant cells, the vacuoles can occupy upto 90% of the volume of the cell
done
clear
B)
Vacuole is bound by a single membrane called tonoplast
done
clear
C)
In Amoeba, the contractile vacuole is important for excretion
done
clear
D)
Plastids are found in all plant cells only
done
clear
View Answer play_arrow
Which of the following is incorrect?
A)
Cell wall of algae is made of cellulose, galactons mannans and minerals like \[CaC{{O}_{3}}\]
done
clear
B)
In plants, cell wall consists of cellulose, hemicellulose, pectins & proteins
done
clear
C)
The middle lamella is a layer mainly of calcium pectate
done
clear
D)
The pili are elongated tubular structures made of a special protein called pilum
done
clear
View Answer play_arrow
Adventitious root system is absent in:
A)
Grass
done
clear
B)
Monstera
done
clear
C)
Banyan tree
done
clear
D)
Mustard plant
done
clear
View Answer play_arrow
The flowers are borne in a basipetal order in:
A)
Cymose
done
clear
B)
Racemose
done
clear
C)
Both
done
clear
D)
Depends on climate
done
clear
View Answer play_arrow
Choose incorrect statement:
A)
A sterile stamen is called staminode
done
clear
B)
Stamens are united into two bundles in pea
done
clear
C)
Carpels are free in lotus
done
clear
D)
Ovules are borne on central axis and septa are absent Argemone
done
clear
View Answer play_arrow
Pollen grains transfer from anther to the stigma of a different plant:
A)
Geitonogamy
done
clear
B)
Autogamy
done
clear
C)
Xenogamy
done
clear
D)
Cleistogamy
done
clear
View Answer play_arrow
Pollen grains are long & ribbon like in:
A)
Sea grasses
done
clear
B)
Vallisneria
done
clear
C)
Hydrilla
done
clear
D)
Oxalis
done
clear
View Answer play_arrow
Select combination of viral disease:
A)
Polio & plague
done
clear
B)
Avian flu and diptheria
done
clear
C)
Chicken pox and Tetanus
done
clear
D)
Measles and polio
done
clear
View Answer play_arrow
The individual of a clone is called:
A)
Chanets
done
clear
B)
Gametes
done
clear
C)
Rametes
done
clear
D)
Tametes
done
clear
View Answer play_arrow
Select incorrect combination:
A)
Micro - injection: Animal cell
done
clear
B)
Biolistics: Plants
done
clear
C)
Lysozyme: Bacteria
done
clear
D)
Stanley cohen & Herbert Boyer: Gel electrophoresis
done
clear
View Answer play_arrow
The enzyme crucial for the immune system to function?
A)
DNAase
done
clear
B)
Adenosine deaminase
done
clear
C)
Nuclease
done
clear
D)
Trophase
done
clear
View Answer play_arrow