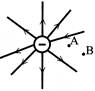
Two charges each \[+\,10\mu C\] of are placed 5 mm apart. Magnitude of electric field at a point I on the axis of the dipole 15 cm away from its centre 0 on the side of the positive charge in N/C:
A)
\[2.9\times {{10}^{6}}\]
done
clear
B)
\[2.6\times {{10}^{6}}\]
done
clear
C)
\[3.5\times {{10}^{6}}\]
done
clear
D)
\[5.12\times {{10}^{6}}\]
done
clear
View Answer play_arrow
Which one of the following is the basic properties of charge?
A)
Quantisation
done
clear
B)
Additivity
done
clear
C)
Conservation
done
clear
D)
All of these
done
clear
View Answer play_arrow
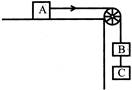
In the given configuration, if a small negative charge moves from B to A then, what is the effect on kinetic energy:
A)
Increase
done
clear
B)
Same
done
clear
C)
Decrease
done
clear
D)
First increase then decrease
done
clear
View Answer play_arrow
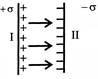
A parallel plate capacitor consists of two large plane parallel plates separated by small distance. What is magnitude of electric field in region I:
A)
Zero
done
clear
B)
\[\frac{Q}{A{{\varepsilon }_{0}}}\]
done
clear
C)
\[\frac{Q}{2A{{\varepsilon }_{0}}}\]
done
clear
D)
\[\frac{3\,Q}{2A{{\varepsilon }_{0}}}\]
done
clear
View Answer play_arrow
A storage battery of emf 8V and internal resistance 0.5V, is being charged by a 120 V dc supply using a series resistor of 15.50. What is the terminal voltage of the battery?
A)
6.5V
done
clear
B)
11.5V
done
clear
C)
9.5V
done
clear
D)
7.5V
done
clear
View Answer play_arrow
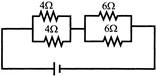
Four resistances are connected in circuit in the given figure. The electric current flowing through 40 and 60 resistance is respectively
A)
\[2A,\text{ }4A~\]
done
clear
B)
1A, 2A
done
clear
C)
1A, 1A
done
clear
D)
2A, 2A
done
clear
View Answer play_arrow
A wire has resistance 120. It is bent in the form of a circle. The effective resistance between the two points on any diameter is equal to:
A)
\[3\Omega \]
done
clear
B)
\[120\]
done
clear
C)
\[60\]
done
clear
D)
\[240\]
done
clear
View Answer play_arrow
Two wires of the same dimensions but resistivities \[{{\rho }_{1}}\] and \[{{\rho }_{2}}\] connected in series. The equivalent resistivity of the combination is:
A)
\[{{\rho }_{1}}+{{\rho }_{2}}\]
done
clear
B)
\[\frac{{{\rho }_{1}}+{{\rho }_{2}}}{2}\]
done
clear
C)
\[2\,({{\rho }_{1}}+{{\rho }_{2}})\]
done
clear
D)
\[\sqrt{{{\rho }_{1}}{{\rho }_{2}}}\]
done
clear
View Answer play_arrow
Circular coil of wire consisting of 100 turns, each of radius 8 cm carries a current of 0.4A. Magnitude of the magnetic field at the centre of the coil:
A)
\[3.1\times {{10}^{-4}}T\]
done
clear
B)
\[2.4\times {{10}^{-\,3}}T\]
done
clear
C)
\[1.36\times {{10}^{-\,3}}T\]
done
clear
D)
\[1.44\times {{10}^{-\,3}}T\]
done
clear
View Answer play_arrow
A long straight wire carries a current of 35A. Magnetic field at a point 20cm from the wire is:
A)
\[5.2\times {{10}^{-5}}T\]
done
clear
B)
\[53.5\times {{10}^{-\,5}}T\]
done
clear
C)
\[26.\times {{10}^{-\,5}}T\]
done
clear
D)
\[1.6\times {{10}^{-\,5}}T\]
done
clear
View Answer play_arrow
Which one of the following has resultant magnetic moment equal to zero?
A)
Paramagnetic
done
clear
B)
Ferromagnetic
done
clear
C)
Diamagnetic
done
clear
D)
Ferrimagnetic
done
clear
View Answer play_arrow
A magnet of magnetic moment M is situated with its axis along the direction of a magnetic field of strength B. The work done in rotating it by an angle of \[180{}^\circ \] will be:
A)
? MB
done
clear
B)
+ MB
done
clear
C)
Zero
done
clear
D)
+ 2MB
done
clear
View Answer play_arrow
The dimension of magnetic flux is
A)
\[MLT{{\,}^{-\,2}}{{A}^{-2}}\]
done
clear
B)
\[MLT{{\,}^{2}}{{A}^{-\,2}}{{A}^{-2}}\]
done
clear
C)
\[MLT{{\,}^{2}}{{A}^{-\,1}}{{A}^{-2}}\]
done
clear
D)
\[MLT{{\,}^{2}}{{A}^{-\,2}}{{A}^{-1}}\]
done
clear
View Answer play_arrow
Magnetic flux linked with a coil, in Wb, is given by the equation then the magnitude of induced emf at t = 2 sec will be:
A)
16V
done
clear
B)
4V
done
clear
C)
8V
done
clear
D)
2V
done
clear
View Answer play_arrow
An ideal coil of 10H is joined in series with a resistance of 5n and a battery of 5V. 2 sec after joining, the current flowing in ampere in the circuit will be:
A)
\[1-e\]
done
clear
B)
\[{{e}^{-1}}\]
done
clear
C)
\[1-\frac{1}{e}\]
done
clear
D)
\[e\]
done
clear
View Answer play_arrow
An alternating voltage is connected in series with a resistance R and an inductance L. If the potential drop across the resistance is 200V and across the inductance is 150V, then voltage applied is:
A)
350V
done
clear
B)
250V
done
clear
C)
300V
done
clear
D)
500V
done
clear
View Answer play_arrow
The impedance of a circuit consists of 3n resistance and \[4\Omega \] resistance. The power factor of the circuit is:
A)
0.4
done
clear
B)
1
done
clear
C)
0.6
done
clear
D)
0.8
done
clear
View Answer play_arrow
When light wave suffers reflection at the interface from air to glass, the change in phase of the reflected wave is equal to:
A)
\[Zero\]
done
clear
B)
\[\frac{\pi }{2}\]
done
clear
C)
\[\pi \]
done
clear
D)
\[2\pi \]
done
clear
View Answer play_arrow
A convex mirror of focal length T forms an image which is \[\frac{1}{n}\] times the object. The distance of the object from the mirror is:
A)
\[(n-1)f\]
done
clear
B)
\[\frac{(n-1)}{n}f\]
done
clear
C)
\[\frac{(n+1)}{n}f\]
done
clear
D)
\[(n+1)f\]
done
clear
View Answer play_arrow
If two waves represented by \[{{y}_{1}}=4\,Sin\,\omega t\] and \[{{y}_{2}}=3Sin\,\left( \omega t+\frac{\pi }{3} \right)\] interfere at a point, the amplitude of the resulting wave is:
A)
7
done
clear
B)
5
done
clear
C)
6
done
clear
D)
3.5
done
clear
View Answer play_arrow
A radioactive substance disintegrates \[{}^{1}/{}_{64}\] of initial value in 60 sec. the half life this substance:
A)
5s
done
clear
B)
10s
done
clear
C)
30s
done
clear
D)
20s
done
clear
View Answer play_arrow
The velocity of an electrons in the first orbit of hydrogen atom is v, then the velocity of an electron in the 2nd orbit of \[H{{e}^{+}}\] is:
A)
\[2v\]
done
clear
B)
\[\frac{v}{2}\]
done
clear
C)
\[v\]
done
clear
D)
\[\frac{v}{4}\]
done
clear
View Answer play_arrow
The ratio of the largest to shortest wavelength in Balmer series of Hydrogen spectra is:
A)
\[\frac{25}{9}\]
done
clear
B)
\[\frac{17}{6}\]
done
clear
C)
\[\frac{9}{5}\]
done
clear
D)
\[\frac{5}{4}\]
done
clear
View Answer play_arrow
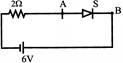
ML The diode shown in the circuit is a silicon diode. The potential difference between the point A and B will be:
A)
6V
done
clear
B)
0.7V
done
clear
C)
0.6V
done
clear
D)
Zero
done
clear
View Answer play_arrow
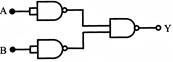
The combination of the gates shown in the figure below produces:
A)
NOR
done
clear
B)
OR
done
clear
C)
AND
done
clear
D)
XOR
done
clear
View Answer play_arrow
Two bodies are thrown up at angles of \[45{}^\circ \] and \[60{}^\circ \] respectively, with the horizontal. If both bodies attain same vertical height, then the ratio of velocities with which these are thrown is:
A)
\[\sqrt{\frac{2}{3}}\]
done
clear
B)
\[\frac{2}{\sqrt{3}}\]
done
clear
C)
\[\frac{\sqrt{3}}{2}\]
done
clear
D)
\[\sqrt{\frac{3}{2}}\]
done
clear
View Answer play_arrow
A ball is thrown upwards from the ground with an initial speed of u. The ball is at a height of 80m at two times, the time interval being 6s. Magnitude of u:
A)
30m/s
done
clear
B)
50m/s
done
clear
C)
20m/s
done
clear
D)
10m/s
done
clear
View Answer play_arrow
A radio wave frequency 840 MHz is sent towards an airoplane. The frequency of the radio echo has a frequency 2.8 KHz more than the original frequency. Then velocity of airoplane is:
A)
3km/s
done
clear
B)
2km/s
done
clear
C)
4km/s
done
clear
D)
0.5km/s
done
clear
View Answer play_arrow
The equation of a transverse wave is given by y \[=20\text{ }Sin\,\pi \,(0.02x-2t)\]where y and x are in cm and t in sec. The wavelength in cm will be:
A)
50
done
clear
B)
200
done
clear
C)
5
done
clear
D)
100
done
clear
View Answer play_arrow
A body cools from \[80{}^\circ \,C\] to \[70{}^\circ \,C\]in 6 minutes. Under identical external condition to cool from \[60{}^\circ \,C\] to\[50{}^\circ C\], it will take:
A)
< 6 minutes
done
clear
B)
6 minutes
done
clear
C)
> 6 minutes
done
clear
D)
None
done
clear
View Answer play_arrow
Stationary waves are set up in an air column. If velocity of sound in air is 330 m/s and frequency is 165 Hz, the distance between the nodes is:
A)
2m
done
clear
B)
0.5m
done
clear
C)
1m
done
clear
D)
4m
done
clear
View Answer play_arrow
Number of beats heard for the super position of \[y=a\text{ }Sin\,3207\pi t\]and \[y=a\text{ }Sin\text{ }326\pi t\]is:
A)
6
done
clear
B)
12
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Frequency of the first overtone of a closed pipe of length \[{{\ell }_{c}}\] is equal to that of the second overtone of an open pipe of length\[{{\ell }_{0}}\]. Then the ratio \[{{\ell }_{c}}/{{\ell }_{o}}\] is equal to:
A)
1/2
done
clear
B)
3
done
clear
C)
1
done
clear
D)
2
done
clear
View Answer play_arrow
The disc of a siren has n holes and the frequency of its rotation is 300 rpm. It produces a note of wavelength 2.4m, when the velocity of sound in air is 360m/s. Then value of n is:
A)
36
done
clear
B)
30
done
clear
C)
5
done
clear
D)
24
done
clear
View Answer play_arrow
If a three block each of mass 4 kg. The table is smooth, the pulley is light and smooth, the string is light and inextensible. The tension in the string connecting B and C is:
A)
4g
done
clear
B)
4g/3
done
clear
C)
4g/3
done
clear
D)
8g/3
done
clear
View Answer play_arrow
A body moving along a straight line with uniform acceleration 'a' it covers a distance \[{{S}_{1}}\] in the first t seconds and a distance \[{{S}_{2}}\] in the next t seconds, a is then given by:
A)
\[\frac{{{S}_{1}}+{{S}_{2}}}{{{t}^{2}}}\]
done
clear
B)
\[\frac{3{{S}_{1}}+{{S}_{2}}}{{{t}^{2}}}\]
done
clear
C)
\[\frac{{{S}_{2}}+{{S}_{1}}}{{{t}^{2}}}\]
done
clear
D)
\[\frac{3{{S}_{1}}+{{S}_{2}}}{2t}\]
done
clear
View Answer play_arrow
A vessel has a height of 50 cm. What is the maximum distance at which water can fall from a hole on its side:
A)
25 cm
done
clear
B)
40 cm
done
clear
C)
33.3 cm
done
clear
D)
50 cm
done
clear
View Answer play_arrow
Acceleration due to gravity become \[\frac{g}{2}\][where g = acceleration due to gravity on the surface of the earth] at a height equal to:
A)
\[4R\]
done
clear
B)
\[\frac{R}{4}\]
done
clear
C)
\[\frac{R}{2}\]
done
clear
D)
\[2R\]
done
clear
View Answer play_arrow
During an adiabatic process, the cube of the pressure is found to be the inversely proportional to the fourth power of the volume. Then the ratio of specific heat is:
A)
1
done
clear
B)
1.33
done
clear
C)
1.67
done
clear
D)
1.4
done
clear
View Answer play_arrow
A shell of mass 20 kg at rest explodes into two fragments whose masses are in the ratio 2 : 3. The smaller fragment moves with a velocity of\[6\text{ }m{{s}^{-1}}\]. The kinetic energy of larger fragment will be:
A)
96 J
done
clear
B)
216 J
done
clear
C)
149 J
done
clear
D)
360 J
done
clear
View Answer play_arrow
Two identical rods AC and CB made of two different metals having thermal conductivities in the ratio 2 : 3 are kept in contact with each other at the end C as shown. A is at \[100{}^\circ C\]and B is at\[25{}^\circ C\]. Then the junction C is at:
A)
\[55{}^\circ C~\]
done
clear
B)
\[60{}^\circ C~\]
done
clear
C)
\[75{}^\circ C\]
done
clear
D)
\[50{}^\circ C\]
done
clear
View Answer play_arrow
A body of mass 10 kg moves at a constant speed of 10 m/s. A constant force then acts for 4 sec on the body and gives it a speed of 2 m/s in opposite direction. Magnitude of the force acting on the body is:
A)
30 N
done
clear
B)
20 N
done
clear
C)
15 N
done
clear
D)
110 N
done
clear
View Answer play_arrow
A 130 m long train is moving in upward direction with speed 72 km/hr. Another train of 120 m long is moving in downward direction with speed of 108 km/hr. The time in which second train crosses the first train will be?
A)
5s
done
clear
B)
10s
done
clear
C)
12s
done
clear
D)
15 s
done
clear
View Answer play_arrow
A string is wound around the rim of a mounted flywheel of mass 20 kg and radius 20 cm. A steady pull of 25 N is applied on the cord (Neglecting friction and mass of string) the angular acceleration of the wheel is:
A)
\[50\text{ }{{s}^{-2}}~\]
done
clear
B)
\[25\text{ }{{s}^{-2}}~\]
done
clear
C)
\[12.5\text{ }{{s}^{-2~}}\]
done
clear
D)
\[6.25\text{ }{{s}^{-2}}\]
done
clear
View Answer play_arrow
What is the radius of curvature of a double convex lens made of glass of refractive index 1.55. If its focal length is 20 cm:
A)
22 cm
done
clear
B)
25cm
done
clear
C)
30cm
done
clear
D)
30cm
done
clear
View Answer play_arrow
The law of definite proportion is not applicable to nitrogen oxide because:
A)
Nitrogen atomic weight is not constant
done
clear
B)
Nitrogen molecular weight is variable
done
clear
C)
Nitrogen equivalent weight is variable
done
clear
D)
Oxygen atomic weight is variable
done
clear
View Answer play_arrow
The hydride ions (H-) are isoelectronic with:
A)
\[Li\]
done
clear
B)
\[H{{e}^{+}}\]
done
clear
C)
\[He~\]
done
clear
D)
\[Be\]
done
clear
View Answer play_arrow
The magnetic quantum number specifies:
A)
Size of orbitals
done
clear
B)
Shape of orbitals
done
clear
C)
Orientation of orbitals
done
clear
D)
Nuclear stability
done
clear
View Answer play_arrow
Chloride of an element a gives neutral solution in water. The element in the periodic table belongs to:
A)
1th group
done
clear
B)
3rd group
done
clear
C)
5th group
done
clear
D)
1st transition series
done
clear
View Answer play_arrow
If the atomic no. of element X is 7. The best electron dot symbol for the element is:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
o-nitrophenol is more volatile than p-nitrophenol due to:
A)
Intramolecular H-bonding
done
clear
B)
Intemolecuair H-bonding
done
clear
C)
Resonance
done
clear
D)
Inductive effect
done
clear
View Answer play_arrow
The r.m.s velocity of hydrogen is \[\sqrt{7}\] times the r.m.s velocity of nitrogen. If T is the temperature of the gas, then:
A)
\[T\,({{H}_{2}})=T({{N}_{2}})\]
done
clear
B)
\[T\,({{H}_{2}})>T({{N}_{2}})\]
done
clear
C)
\[T\,({{H}_{2}})<T({{N}_{2}})\]
done
clear
D)
\[T\,({{H}_{2}})=\sqrt{7}({{N}_{2}})\]
done
clear
View Answer play_arrow
The resultant heat change in a reaction is the same whether it takes place in one or several stages". This statement is called:
A)
Lavoisier and laplaces low
done
clear
B)
Hess's Law
done
clear
C)
Joule's Law
done
clear
D)
Le-chatolier's principle
done
clear
View Answer play_arrow
It the equilibrium constant for the reaction, \[{{N}_{2}}+3{{H}_{2}}2N{{H}_{3}}\]then the equilibrium constant for the reaction \[2{{N}_{2}}+6{{H}_{2}}4N{{H}_{3}}\] would be equal to:
A)
\[{{K}^{2}}\]
done
clear
B)
\[\sqrt{k}\]
done
clear
C)
\[\frac{1}{\sqrt{k}}\]
done
clear
D)
\[\frac{1}{{{k}^{2}}}\]
done
clear
View Answer play_arrow
The pH of a soft drink is 3.82. Its hydrogen ion concentration will be:
A)
\[1.96\times {{10}^{-2}}\,mol\,{{L}^{-1}}\]
done
clear
B)
\[1.96\times {{10}^{-3}}\,mol\,{{L}^{-1}}\]
done
clear
C)
\[1.5\times {{10}^{-4}}\,mol\,{{L}^{-1}}\]
done
clear
D)
\[1.5\times {{10}^{-\,5}}\,mol\,{{L}^{-1}}\]
done
clear
View Answer play_arrow
When \[KMn{{O}_{4}}\] acts as an oxidizing agent and ultimately forms \[{{[Mn{{O}_{4}}]}^{-2}}\],\[Mn{{O}_{3}}\],\[Mn{{O}^{+2}}\] then the number of electrons transferred in each case respectively are:
A)
4, 3, 1, 5
done
clear
B)
1, 5, 3, 7
done
clear
C)
1, 3, 4, 5
done
clear
D)
3, 5, 7, 1
done
clear
View Answer play_arrow
When same amount of zinc is treated separately with excess of Sulphuric acid and excess of sodium hydroxide, the ratio of volumes of hydrogen evolved is:
A)
1 : 1
done
clear
B)
1 : 2
done
clear
C)
2 : 1
done
clear
D)
9 : 4
done
clear
View Answer play_arrow
Which is most basic in character?
A)
CsOH
done
clear
B)
KOH
done
clear
C)
NaOH
done
clear
D)
LiOH
done
clear
View Answer play_arrow
Carborundum is:
A)
SiC
done
clear
B)
PbC
done
clear
C)
CaC
done
clear
D)
SnC
done
clear
View Answer play_arrow
All bonds in benzene are equal due to:
A)
Tautomerism
done
clear
B)
Inductive effect
done
clear
C)
Resance
done
clear
D)
Isomerism
done
clear
View Answer play_arrow
In the formation of methane molecule, carbon makes use of:
A)
\[sp\]- hybridized orbitals
done
clear
B)
\[s{{p}^{3}}\]- hybridized orbitals
done
clear
C)
\[s{{p}^{3}}\] - hybridized orbitals
done
clear
D)
Unhybridized orbitals
done
clear
View Answer play_arrow
Which of the following does not react with methane under normal condition?
A)
\[{{l}_{2}}\]
done
clear
B)
\[C{{l}_{2}}\]
done
clear
C)
\[B{{r}_{2}}\]
done
clear
D)
\[{{F}_{2}}\]
done
clear
View Answer play_arrow
Baeyer's reagent is:
A)
Alkaine \[KMn{{O}_{4}}\]solution
done
clear
B)
Acidic \[KMn{{O}_{4}}\] solution
done
clear
C)
Neutral \[KMn{{O}_{4}}\] solution
done
clear
D)
Aqueous bromine solution
done
clear
View Answer play_arrow
Which of the following decolourize\[KMn{{O}_{4}}\] (Neutral or slightly alkaline)?
A)
Ethane
done
clear
B)
Ethene
done
clear
C)
Acetylene
done
clear
D)
Both B and C
done
clear
View Answer play_arrow
The reaction of benzene with chlorine in the presence of \[FeC{{l}_{3}}\] gives:
A)
Benzene hexachloride
done
clear
B)
Chlorobenzene
done
clear
C)
Benzyl chloride
done
clear
D)
Benzoly chloride
done
clear
View Answer play_arrow
A lake with an inflow of domestic sewage rich in organic waste may result in:
A)
Drying of the lake very soon due to algal bloom
done
clear
B)
An increase production of fish due to lot of nutrients
done
clear
C)
Death of fist due to lack of oxygen
done
clear
D)
Increased population of aquatic food used organisms
done
clear
View Answer play_arrow
The ratio of cationic radius to anionic radius in an ionic crystal is greater than 0.732. Its coordination number is:
A)
6
done
clear
B)
1
done
clear
C)
8
done
clear
D)
4
done
clear
View Answer play_arrow
The ratio of the value of any colligative property for KCl solution to that for sugar solution is nearly:
A)
1
done
clear
B)
0.5
done
clear
C)
2.0
done
clear
D)
3
done
clear
View Answer play_arrow
Beryllium is placed above magnesium in the second group. Beryllium dust, therefore when a added to \[MgC{{l}_{2}}\] solution will:
A)
Have no effect
done
clear
B)
Precipitate Mg metal
done
clear
C)
Precipitate MgO
done
clear
D)
Lead to dissolution of Be metal
done
clear
View Answer play_arrow
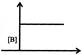
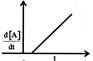
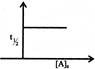
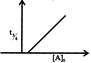
Which graph represents zero - order reaction \[[A(g)\,\to B(g)]\]:
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following electrolytes is least effective in causing flocculation of ferric hydroxide sol:
A)
\[{{K}_{4}}[Fe\,{{(CN)}_{6}}]\]
done
clear
B)
\[{{K}_{2}}Cr{{O}_{4}}\]
done
clear
C)
\[KBr\]
done
clear
D)
\[{{K}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
Zone refining is a technique used primarily for which one of the following process:
A)
Alloying
done
clear
B)
Tempering
done
clear
C)
Sintering
done
clear
D)
Purification
done
clear
View Answer play_arrow
The correct ordect order of solubility in water for He, Ne, Ar, Kr, Xe, is:
A)
He > Ne > Ar > Kr > Xe
done
clear
B)
Ne > Ar > Kr > He > Xe
done
clear
C)
Xe > Kr > Ar > Ne > He
done
clear
D)
Ar > Ne > He > Kr > Xe
done
clear
View Answer play_arrow
Which of the following is the correct sequence of atomic weights of given elements?
A)
Fe > Co > N
done
clear
B)
Co > N > Fe
done
clear
C)
Ni > Co > Fe
done
clear
D)
Fe > Ni > Co
done
clear
View Answer play_arrow
Cuprammonium ion \[{{[Cu{{(N{{H}_{3}})}_{4}}]}^{2+}}\] is:
A)
Tetrahedral
done
clear
B)
Square planar
done
clear
C)
Triangular bipyramid
done
clear
D)
Octahedral
done
clear
View Answer play_arrow
The oxidation number of Cr in \[[Cr\,{{(N{{H}_{3}})}_{6}}]C{{l}_{3}}\] is:
A)
8
done
clear
B)
6
done
clear
C)
4
done
clear
D)
3
done
clear
View Answer play_arrow
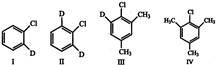
Which of the following will not undergo nucleophile substitution?
A)
II, III and IV
done
clear
B)
II and IV
done
clear
C)
III and IV
done
clear
D)
Only IV
done
clear
View Answer play_arrow
Phenol is less acidic than:
A)
Ethanol
done
clear
B)
o-nitraphenol
done
clear
C)
Methanol
done
clear
D)
p-methylphenol
done
clear
View Answer play_arrow
Paraldehyde is:
A)
A trimer of formaldehyde
done
clear
B)
A trimer of acetaldehyde
done
clear
C)
A hexamer of formaldehyde
done
clear
D)
A hevamer of acetaldehyde
done
clear
View Answer play_arrow
Which of the following is the strongest base?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
An isocyanide on hydrolysis gives:
A)
Amide
done
clear
B)
Carboxylic acid and ammonia
done
clear
C)
N-substituted amide
done
clear
D)
\[1{}^\circ -\]amine and formic acid
done
clear
View Answer play_arrow
Toilet soab is:
A)
A mixture of calcium and sodium salts of higher fatty acids
done
clear
B)
A mixture of potassium stearate and glycerol
done
clear
C)
A mixture of sodium salts of higher fetty acids
done
clear
D)
A mixture of potassium salts of higher felty acids
done
clear
View Answer play_arrow
Which one among the following is a thermosetting plastic?
A)
PVC
done
clear
B)
PVA
done
clear
C)
Bakelite
done
clear
D)
Perspex
done
clear
View Answer play_arrow
Antiseptic chloroxylenol is:
A)
3-chloro-4, 5-dimethylphenol
done
clear
B)
4-chloro-3, 5-dimethylphenol
done
clear
C)
5-chloro-3, 4-dimethyphenol
done
clear
D)
4-chloro-2, 5-dimethyphenol
done
clear
View Answer play_arrow
AgCl dissolves in ammonia solution giving:
A)
\[Ag_{2}^{+}\text{, }NH_{4}^{+}\text{ }and\text{ }C{{l}^{-}}\]
done
clear
B)
\[Ag\,{{\text{(}N{{H}_{3}})}^{+}}\text{ }and\text{ }C{{l}^{-}}\]
done
clear
C)
\[A{{g}_{2}}{{\text{(}N{{H}_{3}})}^{+}}\,and\text{ }C{{l}^{-}}\]
done
clear
D)
\[Ag\,(N{{H}_{3}})\text{ }and\text{ }C{{l}^{-}}\]
done
clear
View Answer play_arrow
0.53g of \[N{{a}_{2}}C{{O}_{3}}\] has been dissolved in 100 ml of a sodium carbonate solution. The normality of the solution:
A)
\[\frac{N}{5}\]
done
clear
B)
\[\frac{N}{2}\]
done
clear
C)
\[\frac{N}{10}\]
done
clear
D)
\[N\]
done
clear
View Answer play_arrow
Which one of the following is expected to exhibit optical isomeris? (en = ethylenediamine):
A)
\[Cis-[Ft{{(N{{H}_{3}})}_{2}}C{{l}_{2}}]\]
done
clear
B)
\[trans\,-[it\,{{(N{{H}_{3}})}_{2}}C{{l}_{2}}]\]
done
clear
C)
\[Cis-{{[CO{{(en)}_{2}}C{{l}_{2}}]}^{+}}\]
done
clear
D)
\[trans\,-{{[CO{{(en)}_{2}}C{{l}_{2}}]}^{+}}\]
done
clear
View Answer play_arrow
A strong base can abstract an a-hydrogen from:
A)
Ketone
done
clear
B)
Alkane
done
clear
C)
Alkene
done
clear
D)
Amine
done
clear
View Answer play_arrow
lodoform test is not given by:
A)
Ethanol
done
clear
B)
Ethanol
done
clear
C)
2-pentanone
done
clear
D)
3-pentanone
done
clear
View Answer play_arrow
Amides may be converted into amines by a reaction named after?
A)
Hoffmann
done
clear
B)
Claisen
done
clear
C)
Perkin
done
clear
D)
Kekule
done
clear
View Answer play_arrow
Which is the first step of taxonomy?
A)
Nomenclature
done
clear
B)
Identification
done
clear
C)
Classification
done
clear
D)
Hierarchical arrangement
done
clear
View Answer play_arrow
Which is the most effective way to conserve the plant diversity of an area?
A)
By creating botanical garden
done
clear
B)
By developing seed bank
done
clear
C)
By creating biosphere reserve
done
clear
D)
By tissue culture method
done
clear
View Answer play_arrow
Who prepare red list for endangered species?
A)
Botanical survey of India
done
clear
B)
Geologica survey of India
done
clear
C)
Zoological survey of India
done
clear
D)
All of the above
done
clear
View Answer play_arrow
The zoological parks are working under supervision of______
A)
CZA
done
clear
B)
IABG
done
clear
C)
ICBN
done
clear
D)
ICZN
done
clear
View Answer play_arrow
Who is 'father of Taxonomy' among the following:
A)
Aristole
done
clear
B)
Maheswari
done
clear
C)
Birbal sahani
done
clear
D)
Linnaeus
done
clear
View Answer play_arrow
The study of Algae is called _____
A)
Taxonomy
done
clear
B)
Algology
done
clear
C)
Mycology
done
clear
D)
Lichenology
done
clear
View Answer play_arrow
Cell wall of fungus made up of _____
A)
Protein
done
clear
B)
Carbohydrates
done
clear
C)
Chitin
done
clear
D)
Cellulose
done
clear
View Answer play_arrow
Which animal is umbrella-shaped and free swiming _____
A)
Jelly-fish
done
clear
B)
Aurelia
done
clear
C)
Hydra
done
clear
D)
A & B
done
clear
View Answer play_arrow
Ascaris is found in:
A)
Tissue
done
clear
B)
Alimentary canal
done
clear
C)
Body cavity
done
clear
D)
Lymph nodes
done
clear
View Answer play_arrow
Companion cells are found in:
A)
Phloem
done
clear
B)
Endodermis
done
clear
C)
Xylem
done
clear
D)
Stomata
done
clear
View Answer play_arrow
Radial vascular bundle are found in ____
A)
Flower
done
clear
B)
Leaf
done
clear
C)
Root
done
clear
D)
Stem
done
clear
View Answer play_arrow
Collenchyma shows deposition of ____
A)
Resin
done
clear
B)
Liginin
done
clear
C)
Pectin
done
clear
D)
Suberin
done
clear
View Answer play_arrow
Which tissue is always absent in root?
A)
Sclerenchyma
done
clear
B)
Meristematic
done
clear
C)
Collenchyma
done
clear
D)
Parenchyma
done
clear
View Answer play_arrow
Which cells regulate the function of sieve tube?
A)
Companion cells
done
clear
B)
Guard cells
done
clear
C)
Balliform cells
done
clear
D)
Passage cells
done
clear
View Answer play_arrow
Give examples of elastic bond:
A)
Cartilage
done
clear
B)
Tendon
done
clear
C)
Ligament
done
clear
D)
[b] and [c] both
done
clear
View Answer play_arrow
What is phyllotaxy:
A)
Arrangement of leaves on stem
done
clear
B)
Folding leaf in the bud
done
clear
C)
[a] & [b] Both
done
clear
D)
None of the above
done
clear
View Answer play_arrow
In monocot male gametophyte is:
A)
Nucleus
done
clear
B)
Microspore
done
clear
C)
Tetral
done
clear
D)
Megaspore
done
clear
View Answer play_arrow
The eye of potato called as ____
A)
Axillary bud
done
clear
B)
Accessory bud
done
clear
C)
Opuntia
done
clear
D)
Aspargaus
done
clear
View Answer play_arrow
Phyllode is present in:
A)
Euphorbia
done
clear
B)
Australian acacia
done
clear
C)
Opuntia
done
clear
D)
Asparagus
done
clear
View Answer play_arrow
What is the edible part in coconut?
A)
Endosperm
done
clear
B)
Fruit wall
done
clear
C)
Entire seed
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Parallel venation is a charactgeristic of:
A)
Grasses
done
clear
B)
Parasitic plants
done
clear
C)
Legames
done
clear
D)
Xerophytic plants
done
clear
View Answer play_arrow
Modified stem of ____ protect the plant from graxing animal:
A)
Aloe vera
done
clear
B)
Gloriosa superba
done
clear
C)
Datura festuosa
done
clear
D)
Cariss a carandus
done
clear
View Answer play_arrow
Which of these characters do not belong to compositae?
A)
Syngenesious stamens
done
clear
B)
Ligulate ray flowers
done
clear
C)
Basal ovules
done
clear
D)
Five lobed stigma
done
clear
View Answer play_arrow
What is the name of second segment of walking leg of cockroach?
A)
Femur
done
clear
B)
Coxa
done
clear
C)
Tarsus
done
clear
D)
Trochanter
done
clear
View Answer play_arrow
What is the smallest cell?
A)
Mycoplasm
done
clear
B)
Yeast
done
clear
C)
Bacteria
done
clear
D)
Blue Green Algae
done
clear
View Answer play_arrow
Who mention that the presence of a cell wall is an unique character of the plant cell?
A)
Virchow
done
clear
B)
Schleiden
done
clear
C)
Schwann
done
clear
D)
Robert Hook
done
clear
View Answer play_arrow
Which the following regarding to examples of keto sugar is not correctly?
A)
Fructose
done
clear
B)
Ribulose
done
clear
C)
Ribose sugar
done
clear
D)
Dinydroxy acetose
done
clear
View Answer play_arrow
Lipids are relatively not soluble in ____
A)
Ether
done
clear
B)
Water
done
clear
C)
Benzene
done
clear
D)
Chloroform
done
clear
View Answer play_arrow
The structure of protein can be destroyed by which rays:
A)
Radio waves
done
clear
B)
Ultra violet rays
done
clear
C)
Infrared rays
done
clear
D)
Micro waves
done
clear
View Answer play_arrow
Find out the incorrect pair:
A)
Telophase - Chromatin is observed
done
clear
B)
\[{{G}_{1}}\] phase - New DNA is synthesized
done
clear
C)
Prophase - Nuclear membrane disintegrates
done
clear
D)
Zygote - synapsis
done
clear
View Answer play_arrow
Which elements are readily mobilized in plants?
A)
S, N, Mo
done
clear
B)
K, N, Mo
done
clear
C)
P, S, N
done
clear
D)
S, N, B
done
clear
View Answer play_arrow
Which element is necessary to stabilize ribosomes?
A)
Mn
done
clear
B)
Ni
done
clear
C)
Mo
done
clear
D)
Mg
done
clear
View Answer play_arrow
Calvin cycle represents one of the following phenomenon:
A)
Dark respiration
done
clear
B)
Dark phosphorylation
done
clear
C)
Oxidative carboxylation
done
clear
D)
Reductive catboxylation
done
clear
View Answer play_arrow
Hill reaction takes place:
A)
In the absence of \[C{{O}_{2}}\]
done
clear
B)
In the presence of carbon dioxide
done
clear
C)
In the absence of a suitable electron acceptor
done
clear
D)
None of the above
done
clear
View Answer play_arrow
Which is the product of aerobic respiration?
A)
Malic acid
done
clear
B)
Ethyl alcohol
done
clear
C)
Pyruvic acid
done
clear
D)
Lactic acide
done
clear
View Answer play_arrow
Which of the following is water stress hormone?
A)
Benxyl amino purine
done
clear
B)
2, 4 - dichlorophenoxy acetic acid
done
clear
C)
Ethylene
done
clear
D)
Abscisic acid
done
clear
View Answer play_arrow
What is 2 - 4 D?
A)
Weedicide
done
clear
B)
Rodenticide
done
clear
C)
Wormicide
done
clear
D)
Insecticide
done
clear
View Answer play_arrow
What is the dental formula of teeth (Adult):
A)
2123/2123
done
clear
B)
2103/2103
done
clear
C)
2003/2003
done
clear
D)
2120/2120
done
clear
View Answer play_arrow
Crown of teeth is made up of:
A)
Dentine
done
clear
B)
Enamel
done
clear
C)
Cement
done
clear
D)
Aereolar
done
clear
View Answer play_arrow
Sound production in human is controlled by
A)
Nares
done
clear
B)
Lungs
done
clear
C)
Larynx
done
clear
D)
Pharynx
done
clear
View Answer play_arrow
The metal ion present in haemoglobin is:
A)
Iron
done
clear
B)
Magnesium
done
clear
C)
Copper
done
clear
D)
Zinc
done
clear
View Answer play_arrow
Blood is flowing in pulmonary vein is:
A)
Oxygenated
done
clear
B)
Deoxygenated
done
clear
C)
Mixed
done
clear
D)
None of the above
done
clear
View Answer play_arrow
The wave representing of ventricles diastole is
A)
P
done
clear
B)
Q
done
clear
C)
R
done
clear
D)
T
done
clear
View Answer play_arrow
ADH is secreted by:
A)
Liver
done
clear
B)
Neurohypophysis
done
clear
C)
Kidney
done
clear
D)
JG cells
done
clear
View Answer play_arrow
What is the function of renin?
A)
Degradation of angiotensinogen
done
clear
B)
Stimulation of corpus lutecim
done
clear
C)
To reduce blood pressure
done
clear
D)
Vasodilation
done
clear
View Answer play_arrow
Where do pollen grains germinate?
A)
Anther
done
clear
B)
Style
done
clear
C)
Stigma
done
clear
D)
Pollen tube
done
clear
View Answer play_arrow
How many eggs contains embryo sac:
A)
3 eggs
done
clear
B)
2 eggs
done
clear
C)
1 egg
done
clear
D)
4 eggs
done
clear
View Answer play_arrow
Wind pollination requires that the pollen grains:
A)
Heavy and wet
done
clear
B)
Heavy and non-sticky
done
clear
C)
Light dry
done
clear
D)
Heavy and sticky
done
clear
View Answer play_arrow
What is scutellum:
A)
An endosperm
done
clear
B)
A seed coat
done
clear
C)
An embryo
done
clear
D)
A cotyledon
done
clear
View Answer play_arrow
Which hormone is released from testes?
A)
Testosterone
done
clear
B)
Estrogen
done
clear
C)
Progesterone
done
clear
D)
Relaxin
done
clear
View Answer play_arrow
What is length a vas deference?
A)
45 inch
done
clear
B)
45mm
done
clear
C)
45 cm
done
clear
D)
4.5 meter
done
clear
View Answer play_arrow
What percentage of semen is produced by seminal vasicles?
A)
50%
done
clear
B)
55%
done
clear
C)
60%
done
clear
D)
65%
done
clear
View Answer play_arrow
When did 'Induced abortion Act came into torce by Government of India?
A)
1st April 1971
done
clear
B)
1st April 1972
done
clear
C)
1st April 1973
done
clear
D)
1st April 1974
done
clear
View Answer play_arrow
Give full from of ELISA?
A)
Enzyme linked Immuno Absorbent Assay
done
clear
B)
Enzyme linked Immuno Assay
done
clear
C)
Enzyme live Implantations Assay
done
clear
D)
Enzyme live Immuno Absorbant
done
clear
View Answer play_arrow
This ration 1:1:1:1 shows ______ cross:
A)
Monohybrid cross
done
clear
B)
Dihybrid cross
done
clear
C)
Back cross
done
clear
D)
Dihybrid test cross
done
clear
View Answer play_arrow
Crossing over during meiosis occurs between:
A)
Sister chromatids
done
clear
B)
Non sister chromatids
done
clear
C)
Centromeres
done
clear
D)
Non-homologus chromosomes
done
clear
View Answer play_arrow
Punnet square is used to know:
A)
Outcome of a cross
done
clear
B)
Probable result of a cross
done
clear
C)
Types of gametes
done
clear
D)
Number of gametes
done
clear
View Answer play_arrow
Which factor in nature causes discontinuous variation in a population?
A)
Recombination
done
clear
B)
Shuffling of porental gene
done
clear
C)
Mutation
done
clear
D)
Gene flow
done
clear
View Answer play_arrow
Nucleus of a cell is the site of synthesis of:
A)
DNA
done
clear
B)
m-RNA
done
clear
C)
t-RNA
done
clear
D)
All
done
clear
View Answer play_arrow
Non-sense codon stakes part in:
A)
Formation of unspecified amino acid
done
clear
B)
Terminating message of gene controlled protein synthesis
done
clear
C)
Releasing t-RNA from polynucleotide chain
done
clear
D)
Conversion of sense DNA into non-sense one
done
clear
View Answer play_arrow
Which one is a Ribozyme?
A)
Melicase
done
clear
B)
Pepticyi transferees
done
clear
C)
Ribonuclease - P
done
clear
D)
Both [b] & [c]
done
clear
View Answer play_arrow
Scientific name of Human beings:
A)
Homoerectus
done
clear
B)
Homo habilis
done
clear
C)
Homo sapiens
done
clear
D)
Hominidae
done
clear
View Answer play_arrow
The first organism to be found on a bare rock is a can ____
A)
Moss
done
clear
B)
Alage
done
clear
C)
Lichen
done
clear
D)
Fern
done
clear
View Answer play_arrow
Who performed this famous experiment to prove origen of life?
A)
Oparin and Haldane
done
clear
B)
Spallanzani and Pasteur
done
clear
C)
Urey and Miller
done
clear
D)
Fox and pasteur
done
clear
View Answer play_arrow
Who is known as the Father of modern bee science:
A)
Fabian
done
clear
B)
Kurain
done
clear
C)
Whenson
done
clear
D)
Huber
done
clear
View Answer play_arrow
Callus is obtained during which time in tissue culture?
A)
2 to 3 days
done
clear
B)
2 to 3 months
done
clear
C)
2 to 3 weeks
done
clear
D)
2 to 3 hours
done
clear
View Answer play_arrow
What is the aim of embryo culture?
A)
Sub culturing
done
clear
B)
Uses of 2, 4 - D and Cytokinins
done
clear
C)
Protoplast isolation
done
clear
D)
Growth of plant lets from dormant seeds
done
clear
View Answer play_arrow
Through plant tissue culture growing the cell tissue and organ in culture medium is called: '
A)
Totipotency
done
clear
B)
Stored food
done
clear
C)
Inter specific hybridization
done
clear
D)
Dormancy
done
clear
View Answer play_arrow
Which one is odd?
A)
Cannabinoids
done
clear
B)
Ganja
done
clear
C)
Charas
done
clear
D)
Smack
done
clear
View Answer play_arrow
What is the full foem IARI:
A)
Indian Agriculture Research Institute
done
clear
B)
International Agrochemical Research Institute
done
clear
C)
Indian Agrochemical Research Institute
done
clear
D)
Indonesia Agronomy Research Institute
done
clear
View Answer play_arrow
Which is used to prevent blood clotting in blood vessels?
A)
Steriods
done
clear
B)
Cyclosporin-A
done
clear
C)
Streptokinase
done
clear
D)
Statins
done
clear
View Answer play_arrow
Ti plasmid naturally occurs in:
A)
Agrobacterium
done
clear
B)
Corynebacterium
done
clear
C)
Staphylococcus
done
clear
D)
Vibrio
done
clear
View Answer play_arrow
The plasmid derived from E. coli is:
A)
pBR327
done
clear
B)
pBR322
done
clear
C)
Both [a] and [b]
done
clear
D)
None
done
clear
View Answer play_arrow
What were the earlier sources of Insulin?
A)
Cattle and pig
done
clear
B)
Carnal and pig
done
clear
C)
Cattle and dog
done
clear
D)
Dog and Pig
done
clear
View Answer play_arrow
Earlier which animals were used to test the safety of polio vaccine:
A)
Transgenic rat
done
clear
B)
Transgenic pigs
done
clear
C)
Transgenic mice
done
clear
D)
Transgenic sheep
done
clear
View Answer play_arrow
A plant formed by the combination of Algae and fungi is a pioneer of which type of succession:
A)
Xerosere
done
clear
B)
Hydrosere
done
clear
C)
Mesosere
done
clear
D)
None of these
done
clear
View Answer play_arrow
Water holding capacity of land depends on?
A)
Soil composition
done
clear
B)
Grain size
done
clear
C)
Aggregation of grain
done
clear
D)
All of these
done
clear
View Answer play_arrow
Food component of the grazing food chain:
A)
Decomposer
done
clear
B)
Primary producer
done
clear
C)
Photosynthetic organism
done
clear
D)
Secondary consumers
done
clear
View Answer play_arrow
Which of the following is placed in upper most (highest) level of ecological pyramids:
A)
Herbivores
done
clear
B)
Carnivores
done
clear
C)
Primary & secondary producers
done
clear
D)
Primary & secondary consumer
done
clear
View Answer play_arrow
In ecosystem the source of energy is:
A)
ATP
done
clear
B)
Sun
done
clear
C)
The green plant
done
clear
D)
Sugar
done
clear
View Answer play_arrow
It helps in absorbtion of phosphorus?
A)
Leaves
done
clear
B)
Mycorrhiza
done
clear
C)
Root
done
clear
D)
Stem
done
clear
View Answer play_arrow
Ranthambore national park is situated in:
A)
Maharashtra
done
clear
B)
Rajasthan
done
clear
C)
Gujrat
done
clear
D)
UP
done
clear
View Answer play_arrow
Which animal has become extinct from India
A)
Snow leopard
done
clear
B)
Hippopotamus
done
clear
C)
Wolf
done
clear
D)
Cheetah
done
clear
View Answer play_arrow
Petroleum is a:
A)
Synthetic product
done
clear
B)
Renewable resource
done
clear
C)
Non-renewable resource
done
clear
D)
Inconvenient resource
done
clear
View Answer play_arrow
Which of the following is considered a hot- spot of biodiversity in India?
A)
Western ghats
done
clear
B)
Indo-Gangetic plain
done
clear
C)
Eastern ghats
done
clear
D)
Aravalli hills
done
clear
View Answer play_arrow
The nature of climax community ultimately depends on:
A)
Climate
done
clear
B)
Bed rock
done
clear
C)
Soil organisms
done
clear
D)
Pool of available nutrients
done
clear
View Answer play_arrow
Green - House effect refers to:
A)
Production of cereals
done
clear
B)
Cooling of earth
done
clear
C)
Trapping UV rays
done
clear
D)
Warming of Earth
done
clear
View Answer play_arrow
Which of the following is pollution related disorder?
A)
Fluorosis
done
clear
B)
Leprosy
done
clear
C)
Pneumonicosis
done
clear
D)
Silicosis
done
clear
View Answer play_arrow
Montreal protocol which calls for appropriate action to protect the ozone layer from "Human activities" was passed in the year:
A)
1986
done
clear
B)
1987
done
clear
C)
1988
done
clear
D)
1985
done
clear
View Answer play_arrow
Which one of the following is not a bio indicator of water pollution?
A)
Sludge worms
done
clear
B)
Blood worms
done
clear
C)
Stone filies
done
clear
D)
Sewage fungus
done
clear
View Answer play_arrow