A full wave rectifier circuit along with the input and output are shown in figure.
The contribution from the diode \[{{D}_{2}}\] is/are
A)
A, C, E
done
clear
B)
B, D, F
done
clear
C)
A, C, E
done
clear
D)
A, B, C, D, E, F
done
clear
View Answer play_arrow
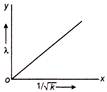
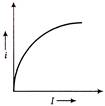
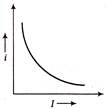
A moving particle has kinetic energy K and wavelength \[\lambda \] then, the correct graph related to the particle is
A)
done
clear
B)
done
clear
C)
Both a and b
done
clear
D)
None of the above
done
clear
View Answer play_arrow
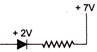
In the network shown below
If current is 5 A and is decreasing at the rate of \[{{10}^{3}}\] A/s, then \[{{V}_{B}}-{{V}_{A}}\] will be
A)
20 V
done
clear
B)
10 V
done
clear
C)
15 V
done
clear
D)
5 V
done
clear
View Answer play_arrow
If one of the slit in Young's double slit experiment is covered with a black opaque paper, then
A)
the fringe width decreases
done
clear
B)
the bright fringes become fainter
done
clear
C)
there will be uniform illumination all over the screen
done
clear
D)
diffraction pattern will be observed
done
clear
View Answer play_arrow
The half-life of a radioactive substance is 20 days. The time taken for \[\frac{3}{4}\] of its original mass to disintegrate is
A)
20 days
done
clear
B)
40 days
done
clear
C)
60 days
done
clear
D)
80 days
done
clear
View Answer play_arrow
If a resistance coil is made by joining in parallel two resistances each of \[10\,\Omega \]. An emf of 1.0 V is applied between two ends of coil for 5 min. The heat produced in calories will be
A)
10.3 cal
done
clear
B)
14.3 cal
done
clear
C)
16.3 cal
done
clear
D)
18.3 cal
done
clear
View Answer play_arrow
If a parallel plate capacitor has capacity C, on inserting a dielectric slab of relative permittivity K and thickness equal to one-fourth of the plate separation is placed between the plates, then its capacity becomes C'. The value of C'/C will be
A)
\[\frac{K}{2\left( K+1 \right)}\]
done
clear
B)
\[\frac{2\,K}{K+1}\]
done
clear
C)
\[\frac{2\,K}{K+1}\]
done
clear
D)
\[\frac{4\,K}{3\,K+1}\]
done
clear
View Answer play_arrow
A body of mass 4 kg is acted upon by a force which causes a displacement in it given by \[\operatorname{x} = {{t}^{2}}\] m, where t is time in second The work done by force in 4s is
A)
64 J
done
clear
B)
128 J
done
clear
C)
200 J
done
clear
D)
240 J
done
clear
View Answer play_arrow
A satellite is a heavily body which revolve around the earth at some height. Suppose a satellite is orbiting around the earth in a circular orbit of radius R from centre of earth then its period of revolution varies as
A)
\[{{R}^{3/2}}\]
done
clear
B)
\[{{R}^{2}}\]
done
clear
C)
R
done
clear
D)
\[\sqrt{R}\]
done
clear
View Answer play_arrow
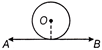
If a thin uniform wire of length X having linear mass density D is bent into a circular loop (as shown in figure). What will be the moment of inertial of the loop about axis AB?
A)
\[\frac{3{{L}^{2}}D}{16\,\pi }\]
done
clear
B)
\[\frac{2{{L}^{2}}D}{3\,\pi }\]
done
clear
C)
\[\frac{3{{L}^{2}}D}{8{{\pi }^{2}}}\]
done
clear
D)
\[\frac{8{{L}^{2}}D}{13{{\pi }^{2}}}\]
done
clear
View Answer play_arrow
If a sphere rolls down from an inclined plane without slipping, then the ratio of translational energy to its total energy is
A)
\[\frac{7}{2}\]
done
clear
B)
\[\frac{2}{7}\]
done
clear
C)
\[\frac{5}{7}\]
done
clear
D)
\[\frac{7}{5}\]
done
clear
View Answer play_arrow
If a wire is suspended vertically from one of its end is stretched by attaching a weight of 200 N to the lower en [d] It stretches the wire by 1 mm, then the potential energy stored in the wire is
A)
1 J
done
clear
B)
0.1 J
done
clear
C)
0.01 J
done
clear
D)
0.001 J
done
clear
View Answer play_arrow
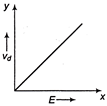
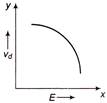
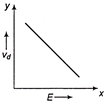
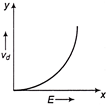
If E denotes electric field in a uniform conductor, I is corresponding current through it and \[{{v}_{d}}\] is drift velocity of \[{{e}^{-}}\], then the correct graph is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
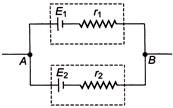
Two batteries of emf \[{{E}_{1}}\,and\,\,{{E}_{2}}\,({{E}_{2}}>{{E}_{1}})\] and internal resistance \[{{r}_{1}}\,\,and\,\,{{r}_{2}}\] respectively are connected in parallel as shown in figure.
A)
The equivalent emf E is smaller than \[{{E}_{1}}\]
done
clear
B)
The equivalent emf \[\operatorname{E} = {{E}_{1}} + {{E}_{2}}\]
done
clear
C)
The equivalent emf E is smaller than \[{{E}_{1}}\]
done
clear
D)
The equivalent emf \[{{E}_{eq}}\] of two cell is between \[{{E}_{1}}\,\,and\,\,{{E}_{2}}\] always
done
clear
View Answer play_arrow
A rod of mass m and length l is lying on a horizontal table. The work done to make it stand on one end will be
A)
2 mgl
done
clear
B)
mgl
done
clear
C)
\[\frac{mgl}{2}\]
done
clear
D)
\[\frac{mgl}{4}\]
done
clear
View Answer play_arrow
Three lenses of focal length \[+15 cm, +150 cm and + 250 cm\] are available, for making an astronomical telescope. To produced the largest magnification, the focal length of the eye-piece should be
A)
+15 cm
done
clear
B)
+150 cm
done
clear
C)
+250 cm
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which of the following represent the correct graph between the intensity of incident photons (J) and the photoelectric current?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
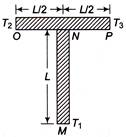
Two identical rods MN and OP, each of length L, cross-sectional area A and thermal conductivity K are connected as shown in figure. Ends M, O and P are maintained at temperatures\[{{T}_{1}} =\,\,20{}^\circ C\], \[{{\operatorname{T}}_{2}}=30{}^\circ C\,\,and\,\,{{T}_{3}}\,\,=\,\,40{}^\circ C\] respectively. The temperature at N is
A)
\[32{}^\circ \text{ }C\]
done
clear
B)
\[33{}^\circ \text{ }C\]
done
clear
C)
\[34{}^\circ \text{ }C\]
done
clear
D)
\[35{}^\circ \text{ }C\]
done
clear
View Answer play_arrow
An electromagnetic wave is propagating in medium with relative magnetic permeability 50 and relative dielectric, constant 2. The wave impedance of such medium is
A)
\[2000\,\,\Omega \]
done
clear
B)
\[1883\,\,\Omega \]
done
clear
C)
\[1550\,\,\Omega \]
done
clear
D)
\[1222\,\,\Omega \]
done
clear
View Answer play_arrow
The distance between the sun and the earth is R. The angular momentum of earth around the sun is proportional to
A)
\[{{R}^{2/3}}\]
done
clear
B)
R
done
clear
C)
\[{{R}^{1/2}}\]
done
clear
D)
\[{{R}^{3/2}}\]
done
clear
View Answer play_arrow
A body is acted by a force F such that \[F\propto \frac{1}{B}\], where B is distance covered by the body then work done by the force in moving body from points \[{{x}_{1}}\,\,and\,\,{{x}_{2}}\] is
A)
\[k(x_{1}^{2}-x_{2}^{2})\]
done
clear
B)
\[k\left( \frac{x_{1}^{2}}{x_{2}^{2}} \right)\]
done
clear
C)
\[k\,ln\left( \frac{x_{2}^{{}}}{x_{1}^{{}}} \right)\]
done
clear
D)
\[k\,ln\left( \frac{x_{2}^{2}}{x_{1}^{2}} \right)\]
done
clear
View Answer play_arrow
A wave motion is described by y\[y\left( x, t \right)=a\,\,sin (kx -\omega t)\]. Then the ratio of the maximum particle velocity to the wave velocity is
A)
\[\frac{\omega }{k}:1\]
done
clear
B)
\[\frac{1}{ka}:1\]
done
clear
C)
\[ka:1\]
done
clear
D)
\[\omega a:1\]
done
clear
View Answer play_arrow
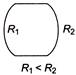
Equal masses of two substances of densities \[{{\rho }_{A}}\,\,and\,\,{{\rho }_{B}}\] are mixed together. The density of the mixture would be
A)
\[\sqrt{{{\rho }_{A}}\,{{\rho }_{B}}}\]
done
clear
B)
\[\frac{{{\rho }_{A}}+\,{{\rho }_{B}}}{2}\]
done
clear
C)
\[\frac{{{\rho }_{A}}\,{{\rho }_{B}}}{{{\rho }_{A}}+\,{{\rho }_{B}}}\]
done
clear
D)
\[\frac{2{{\rho }_{A}}\,{{\rho }_{B}}}{{{\rho }_{A}}+\,{{\rho }_{B}}}\]
done
clear
View Answer play_arrow
Suppose an infinite number of charges equal to q are placed along x-axis at \[\operatorname{x}=1, y= 2\,\,x=4,\,\,x=8......\] so on. The value of electric field at origin will be
A)
\[\frac{q}{2\pi {{\varepsilon }_{0}}}\]
done
clear
B)
\[\frac{q}{3\pi {{\varepsilon }_{0}}}\]
done
clear
C)
\[\frac{q}{4\pi {{\varepsilon }_{0}}}\]
done
clear
D)
\[\frac{q}{8\pi {{\varepsilon }_{0}}}\]
done
clear
View Answer play_arrow
Displacement x of a particle moving in one dimension is related to time t by the equation\[\operatorname{t} =\sqrt{x} +2\]. The displacement of the particle when its velocity is zero, is (Here x is in metre and t in second)
A)
4
done
clear
B)
2
done
clear
C)
1
done
clear
D)
0
done
clear
View Answer play_arrow
In an inductor, the current I varies with time t as\[\operatorname{I} =4+4\,t\]. If the emf induced in the inductor is 8 mV, what is self-inductance? (Here current is in ampere and time is in second)
A)
\[8\times {{10}^{-6}}H\]
done
clear
B)
\[6\times {{10}^{-6}}H\]
done
clear
C)
\[4\times {{10}^{-3}}H\]
done
clear
D)
\[2\times {{10}^{-3}}H\]
done
clear
View Answer play_arrow
If the dimension of a physical quantity is given by \[{{M}^{a}}{{L}^{b}}{{T}^{c}}\], then the physical quantity will be
A)
acceleration, if \[\operatorname{a} = 1,\,\,b = -1,\, c = - 2\]
done
clear
B)
velocity, if \[\operatorname{a} = 1,\,\,b = 0,\, c = -1\]
done
clear
C)
pressure, if \[\operatorname{a} = 1,\,\,b = -1,\, c = -2\]
done
clear
D)
force, if \[\operatorname{a} = 0,\,\,b = -1,\, c = -2\]
done
clear
View Answer play_arrow
A TV tower has a height of 20 m. The maximum distance upto which TV transmission can be received is equal to \[\left( radius of earth = 6.4 \times 1{{0}^{6}}m \right)\]
A)
\[4 \times 1{{0}^{3}}m\,\]
done
clear
B)
\[16 \times 1{{0}^{3}}m\,\]
done
clear
C)
\[32 \times 1{{0}^{6}}m\,\]
done
clear
D)
\[64 \times 1{{0}^{6}}m\,\]
done
clear
View Answer play_arrow
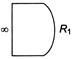
In which of the following case of spherical lenses (as shown in figure), the emergent ray is parallel to incident ray?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A metallic triangle is placed in a uniform electric field [a] In figure which path will the lines of force follow?
A)
1
done
clear
B)
2
done
clear
C)
3
done
clear
D)
4
done
clear
View Answer play_arrow
Two blocks of masses \[{{\operatorname{m}}_{1}}\,=\,\,1\,kg\,\,and\,\,{{m}_{2}}=2\,kg\] are connected by massless string and placed on a horizontal frictionless surface as shown in figure. A force \[\operatorname{F} =\,\,12\,N\] is applied to mass \[{{m}_{1}}\] as shown. The acceleration of the system is
A)
\[12\,m/{{s}^{2}}\]
done
clear
B)
\[8\,m/{{s}^{2}}\]
done
clear
C)
\[4\,m/{{s}^{2}}\]
done
clear
D)
\[2\,\,m/{{s}^{2}}\]
done
clear
View Answer play_arrow
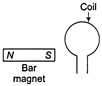
In which case there is no current induced in the coil as shown in figure?
A)
When both moves towards each other
done
clear
B)
When magnet moves towards coil
done
clear
C)
When coil and magnet both move in same direction with same velocity
done
clear
D)
When both moves away from each other with same speed
done
clear
View Answer play_arrow
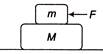
If a block of mass M is lying on a frictionless surface and an another block of mass m is lying on this block as shown in figure. If the coefficient of static friction between two blocks is \[\mu \] then the minimum horizontal force F that must be applied to block of mass m so that it moves over block of mass M is
A)
\[\frac{mg}{\mu }\]
done
clear
B)
mg
done
clear
C)
\[\mu mg\]
done
clear
D)
\[\mu Mg\]
done
clear
View Answer play_arrow
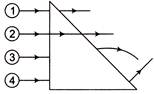
In which of the following figure, the p-n diode is forward biased?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
A positively charged particle enters a magnetic field of value \[B\,\hat{i}\] with a velocity\[v\,\hat{j}\]. The particle will move along
A)
z-axis
done
clear
B)
- z-axis
done
clear
C)
- x-axis
done
clear
D)
- y-axis
done
clear
View Answer play_arrow
A light is an electromagnetic wave consist of particles called photon, if a photon travelling in air enters into a glass slab of large thickness then which of the following quantities will remain unchanged?
A)
velocity
done
clear
B)
wavelength
done
clear
C)
momentum
done
clear
D)
Energy
done
clear
View Answer play_arrow
A vessel whose bottom has round holes with diameter of 1 mm is filled with water. Assuming that surface tension acts only at holes, then maximum height upto which water can be filled in vessel without leakage's (surface tension of water is \[75\times {{10}^{-3}}\,\,N/m,\,\,g\,\,=\,\,10\,\,m/{{s}^{2}})\]
A)
0.03 m
done
clear
B)
0.3 m
done
clear
C)
0.3 cm
done
clear
D)
0.03 cm
done
clear
View Answer play_arrow
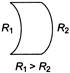
For a streamline flow of water which of the following options is correct
A)
Streamlines may be straight or curved
done
clear
B)
Two streamlines do not intersect each other
done
clear
C)
Only [a] is correct
done
clear
D)
Both [a] and [b] are correct
done
clear
View Answer play_arrow
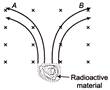
The radiations emitted from a radioactive material separated into two groups A and B when a magnetic field is directed into the plane of the paper. According to figure, names of radiation A and B are respectively
A)
\[\gamma ,\,\,\alpha \]
done
clear
B)
\[\alpha ,\,\,\gamma \]
done
clear
C)
\[\beta ,\,\,\gamma \]
done
clear
D)
\[\alpha ,\,\,\beta \]
done
clear
View Answer play_arrow
Two coherent sources of intensity ratio \[100:1\] undergoes interference, then the ratio of intensities between minima and maxima will be
A)
\[\frac{9}{11}\]
done
clear
B)
\[\frac{3}{4}\]
done
clear
C)
\[\frac{81}{121}\]
done
clear
D)
\[\frac{144}{212}\]
done
clear
View Answer play_arrow
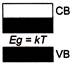
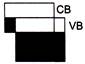
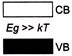
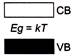
Which one of the energy band diagrams shown in the figure corresponds to that of a semiconductor?
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
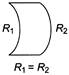
Two springs of spring constant \[{{k}_{1}}\,\,and\,\,{{k}_{\text{2}}}\] have equal maximum velocities, while executing simple harmonic motion. The ratio of their amplitudes will be (masses are equal in both cases)
A)
\[{{\left( \frac{{{k}_{1}}}{{{k}_{2}}} \right)}^{1/2}}\]
done
clear
B)
\[{{\left( \frac{{{k}_{2}}}{{{k}_{1}}} \right)}^{1/2}}\]
done
clear
C)
\[\frac{{{k}_{1}}}{{{k}_{2}}}\]
done
clear
D)
\[\frac{{{k}_{2}}}{{{k}_{1}}}\]
done
clear
View Answer play_arrow
If bullet of mass m and velocity v is fired into a large block of mass M. The final velocity of the system
A)
\[\frac{Mv}{M+m}\]
done
clear
B)
\[\frac{mv}{M+m}\]
done
clear
C)
\[\frac{m}{M}v\]
done
clear
D)
\[\frac{M}{m}v\]
done
clear
View Answer play_arrow
A plane transparent glass slab is placed over various coloured letters, the letter which appears to be raised the least is
A)
green
done
clear
B)
indigo
done
clear
C)
violet
done
clear
D)
red
done
clear
View Answer play_arrow
Two rain drops reach the earth with different terminal velocities having ratio 9:4 then the ratio of their volume is
A)
\[3:2\]
done
clear
B)
\[4:9\]
done
clear
C)
\[9:4\]
done
clear
D)
\[27:8\]
done
clear
View Answer play_arrow
The most acidic species among the following one is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
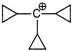
Which of the following has linear geometry?
A)
\[l_{3}^{-}\]
done
clear
B)
\[l_{2}^{+}\]
done
clear
C)
\[{{H}_{2}}O\]
done
clear
D)
\[S{{O}_{2}}\]
done
clear
View Answer play_arrow
If the mass of electron is \[9.11 \times 1{{0}^{-}}^{31}\,kg\], Planck?s constant is \[6.626 \times 1{{0}^{-}}^{34}\,J-s\] and uncertainty m position is \[0.1\,\overset{{}^\circ }{\mathop{A}}\,\], then uncertainty in velocity is
A)
\[5.79\,\,\times \,\,{{10}^{8}}\,m/s\]
done
clear
B)
\[5.79\,\,\times \,\,{{10}^{5}}\,m/s\]
done
clear
C)
\[5.79\,\,\times \,\,{{10}^{6}}\,m/s\]
done
clear
D)
\[5.79\,\,\times \,\,{{10}^{7}}\,m/s\]
done
clear
View Answer play_arrow
An electron, a proton and an alpha particle have KE of 16 E, 4E and E respectively. What is the qualitative order of their de-Broglie wavelengths?
A)
\[{{\lambda }_{e}}>{{\lambda }_{p}}>{{\lambda }_{\alpha }}\]
done
clear
B)
\[{{\lambda }_{p}}={{\lambda }_{\alpha }}>{{\lambda }_{e}}\]
done
clear
C)
\[{{\lambda }_{p}}<{{\lambda }_{e}}<{{\lambda }_{\alpha }}\]
done
clear
D)
\[{{\lambda }_{\alpha }}<{{\lambda }_{e}}={{\lambda }_{p}}\]
done
clear
View Answer play_arrow
Which of the following is not paramagnetic?
A)
\[O_{2}^{2-}\]
done
clear
B)
\[{{B}_{2}}\]
done
clear
C)
\[N_{2}^{+}\]
done
clear
D)
\[O_{2}^{{}}\]
done
clear
View Answer play_arrow
Normality and molarity changes with temperature because they involve volumes. The normality of 0.3 M. phosphoric acid, \[{{H}_{3}}P{{O}_{4}}\] is
A)
0, 9
done
clear
B)
0.1
done
clear
C)
0.3
done
clear
D)
0.6
done
clear
View Answer play_arrow
In a compound, atoms of element Y form CCP lattice and those of element X occupy 2/3rd of tetrahedral voids. The formula of compound will be
A)
\[{{X}_{3}}{{P}_{4}}\]
done
clear
B)
\[{{X}_{4}}{{Y}_{3}}\]
done
clear
C)
\[{{X}_{2}}{{Y}_{3}}\]
done
clear
D)
\[{{X}_{2}}Y\]
done
clear
View Answer play_arrow
The solution which maintains its pH constant even upon addition of small amounts of acid or base, is called buffer solution. Which can act as a buffer?
A)
\[\operatorname{N}{{H}_{4}}Cl\,\,+\,\,N{{H}_{4}}OH\]
done
clear
B)
\[{{\operatorname{CH}}_{3}}COOH\,\,+\,\,C{{H}_{3}}COONa\]
done
clear
C)
40 ml of 0.1 M NaCN + 20 mL of 0.1 M HCN
done
clear
D)
All of the above
done
clear
View Answer play_arrow
Which of the following reaction is not a redox reaction?
A)
\[{{\operatorname{CaCO}}_{3}} \xrightarrow{{}} CaO + C{{O}_{2}}\]
done
clear
B)
\[\operatorname{Na}\,\,+\,\,{{H}_{2}}O\,\,\xrightarrow{{}}\,\,NaOH\,\,+\frac{1}{2}\,{{H}_{2}}\]
done
clear
C)
\[{{\operatorname{MnCl}}_{3}}\,\,\xrightarrow{{}}\,\,MnC{{l}_{2}}\,+\,\,\frac{1}{2}C{{l}_{2}}\]
done
clear
D)
\[{{O}_{2}}+2{{H}_{2}}\,\xrightarrow{{}}\,2{{H}_{2}}O\]
done
clear
View Answer play_arrow
Which of the following has maximum number of unpaired electrons?
A)
\[M{{g}^{2\text{+}}}\]
done
clear
B)
\[T{{i}^{3\text{+}}}\]
done
clear
C)
\[F{{e}^{2\text{+}}}\]
done
clear
D)
\[C{{u}^{2\text{+}}}\]
done
clear
View Answer play_arrow
During electrophilic substitution reaction of fullerene, the hybridisation of carbon atom
A)
remains \[s{{p}^{2}}\] in reactant as well as product
done
clear
B)
changes from \[{{\operatorname{sp}}^{2}}\,to s{{p}^{3}}\]
done
clear
C)
changes from \[s{{p}^{3}}\,to\text{ }s{{p}^{2}}\]
done
clear
D)
cannot be determined
done
clear
View Answer play_arrow
Solids are attracted by magnetic field due to the presence of atoms, ions or molecules with unpaired electron, called paramagnetic. Which among the following is paramagnetic?
A)
\[\operatorname{C}{{l}_{2}}{{O}_{7}}\]
done
clear
B)
\[\operatorname{C}{{l}_{2}}O\]
done
clear
C)
\[\operatorname{C}l{{O}_{2}}\]
done
clear
D)
\[\operatorname{C}{{l}_{2}}{{O}_{5}}\]
done
clear
View Answer play_arrow
Count the total number of \[SO\] bonds, which are having equal length in bisulphate ion?
A)
3
done
clear
B)
4
done
clear
C)
5
done
clear
D)
6
done
clear
View Answer play_arrow
Which of the following statement (s) is/are incorrect about aspirin?
[a] Aspirin is analgesic
[b] Aspirin is antipyretic
[C] Aspirin is tranquilizer
[d] Aspirin doesn't belong to narcotic analgesic
Choose the correct code out of the following.
A)
Only A
done
clear
B)
C and D
done
clear
C)
A and C
done
clear
D)
B and D
done
clear
View Answer play_arrow
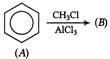
What will be the product when phenol is treated with \[C{{O}_{\text{2}}}\] at \[140-200{}^\circ C\]?
A)
o and p-salicylic acid
done
clear
B)
m and p-salicylic acid
done
clear
C)
o and m-salicylic acid
done
clear
D)
Benzoic acid
done
clear
View Answer play_arrow
Which of the following compound produces acetaldehyde on ozonolysis followed by acidic hydrolysis? [a] [b] [c] \[C{{H}_{3}}C{{H}_{2}}CH=C{{H}_{2}}\] [d] \[C{{H}_{3}}-C\equiv C-C{{H}_{3}}\]
Choose the correct choice out of the following.
A)
A and C
done
clear
B)
A and B
done
clear
C)
A, B and C
done
clear
D)
Only A
done
clear
View Answer play_arrow
What is the intermediate involved in Reimer Tiemann reaction?
A)
Carbocation
done
clear
B)
Carbanion
done
clear
C)
Carbene
done
clear
D)
None of these
done
clear
View Answer play_arrow
What will be the product of the following reaction?
A)
Propanol
done
clear
B)
Propanol
done
clear
C)
Acetaldehyde
done
clear
D)
None of these
done
clear
View Answer play_arrow
What will be the product if product obtained in following reaction undergo oxidation?
A)
Benzyl alcohol
done
clear
B)
Benzoic acid
done
clear
C)
Toluene
done
clear
D)
None of these
done
clear
View Answer play_arrow
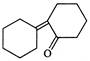
Cyclohexanone on reaction with \[O{{H}^{\Theta }}\] produces.
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Maximum oxidation state exhibited by Mn and its magnetic moment in +2 oxidation state will be
A)
+2, 5.92 BM
done
clear
B)
+7, 4.83 BM
done
clear
C)
+6, 3.87 BM
done
clear
D)
+7, 5.92 BM
done
clear
View Answer play_arrow
Purification of blood can be done by
A)
dialysis
done
clear
B)
filtration
done
clear
C)
electro dialysis
done
clear
D)
coagulation
done
clear
View Answer play_arrow
The oxidation state of Ni in tetra carbonyl nickel is
A)
+1
done
clear
B)
+ 2
done
clear
C)
0
done
clear
D)
+ 4
done
clear
View Answer play_arrow
The effective atomic number of cobalt in \[[Co{{\left( N{{H}_{3}}{{)}_{5}}{{H}_{2}}O \right]}^{3+}}\] is
A)
36
done
clear
B)
33
done
clear
C)
24
done
clear
D)
30
done
clear
View Answer play_arrow
Which of the amino acid contain aromatic side chain?
A)
Histidine
done
clear
B)
Leucine
done
clear
C)
Glycine
done
clear
D)
Valine
done
clear
View Answer play_arrow
Head-to-tail addition takes place in chain growth polymerisation when monomer is
A)
done
clear
B)
\[{{\operatorname{CH}}_{2}}=CH-CH=C{{H}_{2}}~~~\]
done
clear
C)
done
clear
D)
\[C{{H}_{2}}\,=CH\,-C\equiv N\]
done
clear
View Answer play_arrow
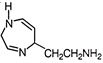
Histamine is an organic nitrogenous compound involved in local immune responsed as well as regulating physiological function in the gut and acting as a neuro transmitter. The durg
is used as
A)
Vasodilator
done
clear
B)
Analgesics
done
clear
C)
Antacid
done
clear
D)
Antiseptic
done
clear
View Answer play_arrow
What is the empirical formula of a compound having \[40\,%\] carbon, \[6.66\,%\] hydrogen and \[53.34\,\,%\] oxygen?
A)
\[{{C}_{2}}{{H}_{2}}O\]
done
clear
B)
\[{{C}_{2}}{{H}_{4}}O\]
done
clear
C)
\[C{{H}_{2}}O\]
done
clear
D)
CHO
done
clear
View Answer play_arrow
Saturated solution of \[KN{{O}_{3}}\] is used to make 'salt bridge' because
A)
velocities of both \[{{K}^{+}}\] and \[NO_{3}^{-}\] are nearly the same
done
clear
B)
velocity of \[{{K}^{+}}\] is greater than that of \[NO_{3}^{-}\]
done
clear
C)
velocity of \[NO_{3}^{-}\] is greater than that of \[{{K}^{+}}\]
done
clear
D)
\[KN{{O}_{3}}\] is highly soluble in water
done
clear
View Answer play_arrow
Molality of aqueous solution of 8.0 M ethanol having density 1.025 g/mL is
A)
12.17
done
clear
B)
24.34
done
clear
C)
10.17
done
clear
D)
14.35
done
clear
View Answer play_arrow
Haemoglobin is the iron containing oxygen transport metal lo protein in the red blood cells of all vertebrates as well as the tissue of some invertebrate. The correct statement in respect of protein haemoglobin is that it
A)
acts as an oxygen carrier in the blood,
done
clear
B)
forms antibodies and offers resistance to diseased
done
clear
C)
functions as a catalyst for biological reactions,
done
clear
D)
maintains blood sugar level.
done
clear
View Answer play_arrow
Which of the following is used as an antifreeze in cars for cooling the engine?
A)
\[10\,%\]% (V/V) ethanol solution in water
done
clear
B)
\[35\,%\]% (V/V) solution of ethylene glycol
done
clear
C)
\[45\,%\]% (V/V) solution of ethylene glycol
done
clear
D)
\[20\,%\]% (V/V) ethanol solution in water
done
clear
View Answer play_arrow
Which of the following has strongest hydrogen bonding?
A)
\[O-H \cdot \cdot \cdot N\]
done
clear
B)
\[F-H \cdot \cdot \cdot F\]
done
clear
C)
\[O-H \cdot \cdot \cdot O\]
done
clear
D)
\[O-H \cdot \cdot \cdot F\]
done
clear
View Answer play_arrow
The unit of 1st and zero order reaction in term of molarity are respectively
A)
\[{{\operatorname{s}}^{-}}^{1}, \,M{{s}^{-}}^{1}\]
done
clear
B)
\[{{\operatorname{s}}^{-}}^{1}, \,M\]
done
clear
C)
\[M{{s}^{-}}^{1}\]
done
clear
D)
\[\operatorname{M}, {{s}^{-}}^{1}\]
done
clear
View Answer play_arrow
Carbocations are stablized by three main structural factors. Such factors are neighboring carbon atoms, neighboring carbon-carbon multiple bonds and neighboring atoms with lone pairs. Most stable among the following carbocations is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Borax, disodium tetra borate is an important boron compound, a mineral, and a salt of boric acid. The number of terminal \[B\text{ }OH\] present in borax is
A)
3
done
clear
B)
4
done
clear
C)
5
done
clear
D)
6
done
clear
View Answer play_arrow
The most reactive substrate towards \[{{S}_{N}}2\] reaction among the following is
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
Which of the following pair of solution is expected to show isotonic at same temperature?
A)
0.2 M urea and 0.2 M KCl
done
clear
B)
0.1 M urea and 0.2 M \[CaC{{l}_{2}}\]
done
clear
C)
0.1 M KCl and 0.1 M \[{{\operatorname{Na}}_{2}}S{{O}_{4}}\]
done
clear
D)
0.1 M \[Ca{{\left( N{{O}_{3}} \right)}_{2}}\] and 0.1 M \[{{\operatorname{Na}}_{2}}S{{O}_{4}}\]
done
clear
View Answer play_arrow
Solubility of
in term of solubility product x\[({{K}_{sp}})\] can be given as
A)
\[{{({{K}_{sp}})}^{1/3}}\]
done
clear
B)
\[{{\left( \frac{{{K}_{sp}}}{2} \right)}^{1/3}}\]
done
clear
C)
\[{{\left( \frac{{{K}_{sp}}}{4} \right)}^{1/3}}\]
done
clear
D)
\[{{\left( \frac{{{K}_{sp}}}{2} \right)}^{1/2}}\]
done
clear
View Answer play_arrow
Molarity of a solution obtained by mixing 800 mL 0.6 M HCl with 200 mL 1 M HCl will be
A)
0.4 M
done
clear
B)
1.6 M
done
clear
C)
0.68 M
done
clear
D)
1.68 M
done
clear
View Answer play_arrow
Product of the following reaction will be
A)
done
clear
B)
done
clear
C)
done
clear
D)
done
clear
View Answer play_arrow
For a reversible reaction \[{{N}_{2}}\,\,+\,\,{{O}_{2}}\rightleftharpoons \,\,2NO\] Activation energy of the backward reaction is lower than that of forward reaction. The slope of k versus 1/T graph will be
A)
zero
done
clear
B)
\[-\frac{H}{2.303\,R}\]
done
clear
C)
\[\frac{H}{2.303\,R}\]
done
clear
D)
\[-\frac{\Delta H}{R}\]
done
clear
View Answer play_arrow
pH of 0.5 M aqueous solution of \[\operatorname{HF}\,\,({{K}_{\alpha }}=2\times 1{{0}^{-\,4}})\] is
A)
2
done
clear
B)
4
done
clear
C)
6
done
clear
D)
10
done
clear
View Answer play_arrow
Insulin production and its action in human body are responsible for the level of diabetes. This compound belongs to which of the following categories?
A)
A co-enzyme
done
clear
B)
A hormone
done
clear
C)
An enzyme
done
clear
D)
An antibiotic
done
clear
View Answer play_arrow
The reaction rate for a reactant or product in a particular reaction is intuitively defined as how fast or slow a reaction takes place. Effect of temperature on reaction rate is given by
A)
Claisen- Clapeyron equation
done
clear
B)
Arrhenius equation
done
clear
C)
Gibbs Helmholtz equation
done
clear
D)
Kirchhoff's equation
done
clear
View Answer play_arrow
What is not correct about Darwinism?
A)
Struggle for existence
done
clear
B)
Within each species, there are variations
done
clear
C)
Overproduction
done
clear
D)
Inheritance of variations to off springs
done
clear
View Answer play_arrow
Which of the chemical used in bonding technique?
A)
Shift's reagent
done
clear
B)
Giemsa
done
clear
C)
Quinacrine mustard
done
clear
D)
Tritiated thymidine
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II [a] Isoalleles 1. Rare crossing over [b] Pseudoalleles 2. \[{{I}^{A1}}\,and\,\,{{I}^{A2}}\] [c] Inhibitor gene 3. Epistatic gene
A)
A-1, B-2, C-3
done
clear
B)
A-3, B-2, C-1
done
clear
C)
A-2, B-1, C-3
done
clear
D)
A-1, B-3, C-2
done
clear
View Answer play_arrow
Which of the following shape graph is obtained by exponential growth?
A)
S-shaped
done
clear
B)
J-shaped
done
clear
C)
Straight line
done
clear
D)
Parallel line
done
clear
View Answer play_arrow
Categorised the following diseases as endotoxic and exotoxic? I. Food poisoning II. Bubonic plaque III. Dysentery IV. Tetanus
A)
I-Endotoxic, II-Exotoxic, III-Endotoxic, IV-Exotoxic
done
clear
B)
I-Exotoxic, II-Exotoxic, III-Endotoxic, IV-Endotoxic
done
clear
C)
I-Exotoxic, II-Endotoxic, III-Endotoxic, IV-Exotoxic
done
clear
D)
I-Exotoxic, II-Endotoxic, III-Endotoxic, IV-Endotoxic
done
clear
View Answer play_arrow
Two or more species which are inhabiting the same or overlapping areas are known as
A)
sympatric species
done
clear
B)
allopatric species
done
clear
C)
sibling species
done
clear
D)
neopatric species
done
clear
View Answer play_arrow
Which of the following is a heterocrine gland? I. Pancreases II. Gonads III. Gastric gland IV. Parathyroid
A)
I, II, III and IV
done
clear
B)
I and II only
done
clear
C)
I, II and III
done
clear
D)
I and IV
done
clear
View Answer play_arrow
Which one of the following option is correct about the active transport?
A)
Active transport releases energy
done
clear
B)
Active transport requires energy
done
clear
C)
Active transport produces ATP
done
clear
D)
Active transport produces a toxic substance
done
clear
View Answer play_arrow
Suppose evolution on earth had occurred in such a way that there are 96 ammo acids instead of 20. DNA has 12 different types of bases and DNA synthesis occurs in the same way as today. The minimum number of bases per DNA codon would be
A)
12
done
clear
B)
2
done
clear
C)
8
done
clear
D)
3
done
clear
View Answer play_arrow
Which phy to hormone has vital inhibitory property with respect to seed germination?
A)
IAA
done
clear
B)
\[G{{A}_{3}}\]
done
clear
C)
ABA
done
clear
D)
2, 4-D
done
clear
View Answer play_arrow
Pick out the wrong statement.
A)
In polygonum type of ovule, the functional haploid megaspore enlarge in size and by means of three successive mitotic divisions, gives rise to an eight-nucleate embryo sac.
done
clear
B)
Sporopollenin makes the exine of spores and pollen grains of plants providing resistance to biodegradation.
done
clear
C)
Pollen kit material is secreted by tapetum
done
clear
D)
Occurance of more than four spores from a spore mother cell is called polyspory.
done
clear
View Answer play_arrow
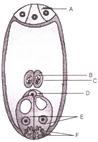
In the given diagram, parts labelled as A, B, C, D,E and F are respectively identified as
A)
A-Polar nuclei B-Egg C-Antipodals D-Filiform apparatus E-Central cell F-Synergids
done
clear
B)
A-Synergids B-Filiform apparatus C-Egg D-Central cell E-Polar nuclei F-Antipodals
done
clear
C)
A-Antipodals B-Polar nuclei C-Central cell D-Egg E-Synergids F-Filiform apparatus
done
clear
D)
A-Polar nuclei B-Central cell C-Egg D-Antipodal E-Filiform apparatus F-Synergids
done
clear
View Answer play_arrow
Which of the following is supposed to be the most direct evidence of organic evolution?
A)
vestigial organ
done
clear
B)
embryos
done
clear
C)
fossils
done
clear
D)
morphology
done
clear
View Answer play_arrow
Match the scientists and their contribution to evolution.
Column I Column II A Charles Darwin 1. Mutation theory B Lamarck 2. Germ plasm theory C Hugo de Vries 3. Origin of species D Ernst Haeckel 4. Philosphie zoologique 5. Biogenetic law
A)
A-1, B-2, C-3, D-4
done
clear
B)
A-3, B-4, C-1, D-5
done
clear
C)
A-5, B-1, C-2, D-3
done
clear
D)
A-2, B-3, C-4, D-5
done
clear
View Answer play_arrow
Which of the following is true regarding ABO blood group?
A)
co-dominance
done
clear
B)
incomplete dominance
done
clear
C)
epistasis
done
clear
D)
multiple alleles
done
clear
View Answer play_arrow
Antibody is produced by antigenic interaction of which of the cells or organ
A)
spleen
done
clear
B)
monocytes
done
clear
C)
lymphocytes
done
clear
D)
leucocytes
done
clear
View Answer play_arrow
In angiosperms, functional megaspore develops as a result of free nuclear division into
A)
embryo sac
done
clear
B)
ovule
done
clear
C)
endosperm
done
clear
D)
zygote
done
clear
View Answer play_arrow
The major role of minor elements inside living organisms is to act as
A)
constituents of hormones
done
clear
B)
binder of cell structure
done
clear
C)
cofactors of enzymes
done
clear
D)
building blocks of important amino acids
done
clear
View Answer play_arrow
An unfair claim to novelty and invention come under
A)
economic piracy
done
clear
B)
resource piracy
done
clear
C)
intellectual piracy
done
clear
D)
theft
done
clear
View Answer play_arrow
A protoxin is
A)
lipid
done
clear
B)
intracellular lipid
done
clear
C)
extracellular crystalline protein
done
clear
D)
a primitive toxin
done
clear
View Answer play_arrow
The Wildlife Protection Act was enacted in
A)
1980
done
clear
B)
1972
done
clear
C)
1969
done
clear
D)
1981
done
clear
View Answer play_arrow
Genetically Modified Organisms (GMO) are developed by rDNA Technology and genetic engineering. They are useful
A)
enhancing food nutritional value
done
clear
B)
reduction in the post-harvest losses
done
clear
C)
increasing the tolerance power of crops against the abiotic stresses
done
clear
D)
All of the above
done
clear
View Answer play_arrow
ADA: Adenosine deaminase enzyme does not function. It occurs as a result of
A)
hormonal defect
done
clear
B)
genetic defect
done
clear
C)
hereditary defect
done
clear
D)
None of these
done
clear
View Answer play_arrow
Which of the following bond is formed during the condensation of mono saccharides?
A)
Covalent bond
done
clear
B)
Glycosidic bond
done
clear
C)
Peptide bond
done
clear
D)
Ionic bond
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A Rabbit 1. Panthera tigris B Tiger 2. Platianista C Ganges Dolphin 3. Oryctolagus D Dog 4. Can is lupus familiaris
Codes
A)
A-1, B-2, C-3, D-4
done
clear
B)
A-1, B-4, C-3, D-2
done
clear
C)
A-3, B-2, C-4, D-1
done
clear
D)
A-2, B-1, C-4, D-3
done
clear
View Answer play_arrow
Provirus differs from prophase in
A)
integration of copy DNA of a retrovirus with host chromosome (DNA)
done
clear
B)
integration of RNA with host DNA
done
clear
C)
integration of genetic DNA with host DNA
done
clear
D)
All of the above
done
clear
View Answer play_arrow
At what age group, measles vaccines should be given to a children?
A)
During pregnancy
done
clear
B)
16 years
done
clear
C)
5-6 years
done
clear
D)
9-15 months
done
clear
View Answer play_arrow
There are two portal system in body. One of them Hypophysial portal system is found in
A)
brain
done
clear
B)
kidney
done
clear
C)
liver
done
clear
D)
heart
done
clear
View Answer play_arrow
Identify whether the given statements are the characteristic of cleavage or typical mitosis. I. DNA synthesis occurs at the normal rate. II. Nuclear/cytoplasmic ratio increases as cleavage progresses. III. Size of daughter cells decreases. IV. Interphase is long
A)
Cleavage-I, II Typical mitosis - III, IV
done
clear
B)
Cleavage-I Typical mitosis - II, III, IV
done
clear
C)
Cleavage-II Typical mitosis -I, III, IV
done
clear
D)
Cleavage-II, III Typical mitosis -I, IV
done
clear
View Answer play_arrow
Addiction of LSD leads to
A)
hallucination
done
clear
B)
damage to kidney
done
clear
C)
mental and emotional disturbance
done
clear
D)
damage to lungs
done
clear
View Answer play_arrow
In frog, the pharynx communicates with tympanic cavity ventrally through
A)
semi-circular canal
done
clear
B)
Bidder's canal
done
clear
C)
Eustachian tube
done
clear
D)
horizontal canal
done
clear
View Answer play_arrow
Which one of the statement is not correct?
A)
The rate of absorption of water is almost directly proportional to the rate of transpiration.
done
clear
B)
Soil temperature, soil aeration, relative humidity, amount of soil water and transpiration are factors affect the absorption.
done
clear
C)
Water is removed in the form of vapours during transpiration.
done
clear
D)
If the atmosphere is humid, it increases the rate of transpiration.
done
clear
View Answer play_arrow
Cystic fibrosis is caused due to
A)
recessive sex-linked gene on X-chromosome
done
clear
B)
recessive autosomal gene on chromosome 7
done
clear
C)
dominant sex-linked gene on X-chromosome
done
clear
D)
dominant autosomal gene on chromosome 11
done
clear
View Answer play_arrow
Identify the state of glandular tissue of mammary glands during the following conditions. I. Non-pregnant woman II. Pregnant woman III. On the infant?s birth IV. After menopause
A)
I-Atrophy II- Influence of oxytocin III- Scanty IV- Influence of oestrogen
done
clear
B)
I-Influence of oestrogen II- Scanty III- Influence of prolactin IV-Atrophy
done
clear
C)
I-Scanty II-Influence of oestrogen and progesterone III-Influence of prolactin IV-Atrophy
done
clear
D)
I-Influence of progesterone II-Influence of prolactin III-Influence of oxytocin IV-Atrophy
done
clear
View Answer play_arrow
Cholesterol and phospholipids concentration is highest in
A)
striated muscle
done
clear
B)
involuntary muscle
done
clear
C)
cardiac muscle
done
clear
D)
smooth muscle
done
clear
View Answer play_arrow
Consider the following statements. I. Green muffler scheme involves the growing green plants along roadside to reduce air pollution. II. Delhi became the first city of the world to use CNG for its public transport system and auto rickshaws by the end of 2002. III. In India, the Air (Prevention and Control of Pollution) Act came into force in 1981 but was amended in 1987 to include noise as an air pollutant. Which of the statements given above are correct?
A)
I and II
done
clear
B)
I and III
done
clear
C)
II and III
done
clear
D)
I, II and III
done
clear
View Answer play_arrow
Motor nerve arising from the floor of midbrain is
A)
hypoglossal
done
clear
B)
abducens
done
clear
C)
pathetic
done
clear
D)
ophthalmic
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II [a] Iron bacteria 1. Carbonydomonas, Bacillus oligocarbophilus [b] Hydrogen bacteria 2. Beggiatoa, Thiothrix [c] Sulphur bacteria 3. Bacillus pentotrophs [d] Carbon bacteria 4. Leptothrix and Cladothrix
Codes
A)
A-4, B-2, C-3, D-1
done
clear
B)
A-3, B-4, C-1, D-2
done
clear
C)
A-4, B-3, C-2, D-1
done
clear
D)
A-2, B-3, C-4, D-1
done
clear
View Answer play_arrow
Consider the following statements. I. Perienth is the collective name of the non-essential floral organs if there is no distinction between sepals and petals. II. The term tepals is used to describe the perianth lobes, which appear like petals. [a] The corolla of Hibiscus is polypetalous and twisted [b] Ovary is superior perigynous flower. Which of the statements given above are correct?
A)
I, II and III
done
clear
B)
I, III and IV
done
clear
C)
II, III and IV
done
clear
D)
All of these
done
clear
View Answer play_arrow
"Nothing in biology makes sense except in the light of evolution" who said this?
A)
Hugo de Vries
done
clear
B)
Charles Darwin
done
clear
C)
Jean Baptiste de Lamarck
done
clear
D)
Theodosius Dobzhansky
done
clear
View Answer play_arrow
Which system or the body grants a patent?
A)
Local body
done
clear
B)
State government
done
clear
C)
Central government
done
clear
D)
Legal system
done
clear
View Answer play_arrow
AIDS symptom appear because it causes
A)
autoimmunity
done
clear
B)
reduction in number of killer T-cells
done
clear
C)
reduction in number of helper T-cells
done
clear
D)
non-production of interferes
done
clear
View Answer play_arrow
Protein coat of a virus enclosing nucleic acid is known as
A)
genome
done
clear
B)
vector
done
clear
C)
plasmid
done
clear
D)
capsid
done
clear
View Answer play_arrow
Morphan's syndrome is caused by
A)
epistatic genes
done
clear
B)
co dominant genes
done
clear
C)
pleiotropic genes
done
clear
D)
polymeric genes
done
clear
View Answer play_arrow
Restriction enzyme cuts DNA at palandronic base sequences which happens to be
A)
TATAGC
done
clear
B)
AAGTTC
done
clear
C)
GTATATC
done
clear
D)
GAATTC
done
clear
View Answer play_arrow
Tetany results due to the deficiency of
A)
insulin
done
clear
B)
epinephrine
done
clear
C)
parathyroid hormone
done
clear
D)
thyroxine
done
clear
View Answer play_arrow
Which one of the statement given below is not correct?
A)
Sulphur is constituent of certain amino acid. The amino acid form the protein by polymerisation.
done
clear
B)
A regular supply of nitrogen to the plants is maintained through nitrogen cycle.
done
clear
C)
Biological nitrogen fixation is the second most important natural process and the major source of nitrogen fixation, which is performed by two types of prokaryotes; bacteria and cyanobacteria
done
clear
D)
Incorporation of nitrogen into amino acid is called ammonification.
done
clear
View Answer play_arrow
Match the following columns.
Column I Column II A Miscarriage 1. Premature degeneration of corpus luteum B Pregnancy test 2. Animal pole C Luteal phase 3. Progesterone secretion D Polar bodies 4. hCG
A)
A-1, B-3, C-2, D-4
done
clear
B)
A-1, B-4, C-3, D-2
done
clear
C)
A-3, B-2, C-4, D-1
done
clear
D)
A-2, B-1, C-4, D-3
done
clear
View Answer play_arrow
How many bones are fused to become the total count of 206?
A)
270
done
clear
B)
250
done
clear
C)
350
done
clear
D)
370
done
clear
View Answer play_arrow
Which of the following is incorrect about grasslands?
A)
Pampas - South America
done
clear
B)
Velds - South Africa
done
clear
C)
Downs - USA
done
clear
D)
Prairies - Canada
done
clear
View Answer play_arrow
PCR uses which of the following?
A)
Primase
done
clear
B)
RNA polymerase
done
clear
C)
Taq polymerase
done
clear
D)
Ligase
done
clear
View Answer play_arrow
Thermo cycler is used in
A)
micro propagation
done
clear
B)
hybridisation
done
clear
C)
PCR
done
clear
D)
fermentation
done
clear
View Answer play_arrow
The infective stage of Plasmodium is found in insect vector is
A)
trophozoite stage
done
clear
B)
amoeboid stage
done
clear
C)
ookinetic stage
done
clear
D)
sporozoite stage
done
clear
View Answer play_arrow
Pick out the wrong statement.
A)
Double fertilisation is unique to gymnosperms and monocotyledons.
done
clear
B)
Egg apparatus of angiosperms consist of two synergids and one egg cell.
done
clear
C)
In porogamy, pollen tube enters the ovule through the micropyle.
done
clear
D)
The ovule attached to the placenta of ovary wall by funicle.
done
clear
View Answer play_arrow
Number of nephrons in a kidney is equal to
A)
the number of Bowman's capsule
done
clear
B)
sum of Bowman's capsule and Malpighi an capsules
done
clear
C)
sum of Bowman's capsule and glomeruli
done
clear
D)
double the number of Bowman's capsule
done
clear
View Answer play_arrow
In which of the following germination will be maximum? \[R = Red light 660 m\mu \] \[\operatorname{Fr} = Far light 730 m\mu \]
A)
\[Seeds + R + Fr + R + Fr\]
done
clear
B)
\[Seeds + R + Fr\]
done
clear
C)
\[Seeds + R + Fr + R~\]
done
clear
D)
\[Seeds + R\]
done
clear
View Answer play_arrow
Shikimic acid can be made from
A)
xylulose
done
clear
B)
erythrose -4- phosphate
done
clear
C)
ribulose
done
clear
D)
None of these
done
clear
View Answer play_arrow
Consider the following statements and choose the correct option given below. I Nepenthes khasiana is a green plant but shows heterotrophic nature for nitrogen supply, therefore, is called as carnivorous. II Frankia is symbiont in root nodules of several legume plants like Pisum sativum.
A)
Statement I is correct, II is incorrect,
done
clear
B)
Statement I is incorrect, II is correct.
done
clear
C)
Both are correct
done
clear
D)
Both are incorrect
done
clear
View Answer play_arrow
Morphogenesis is controlled by
A)
gibberellin and cytokinin
done
clear
B)
auxin and cytokinin
done
clear
C)
auxin and gibnerellin
done
clear
D)
gibberellin and zeatin
done
clear
View Answer play_arrow
Match the item in column I with column II and choose the correct alternatives.
Column I Column II [a] Pneumatophores 1. Heritiera [b] Haustoria 2. Asparagus [c] Prop roots 3. Viscum [d] Tubercular storage roots 4. Screw pine
Codes
A)
A-2, B-4, C-1, D-3
done
clear
B)
A-2, B-3, C-4, D-1
done
clear
C)
A-4, B-1, C-2, D-3
done
clear
D)
A-3, B-2, C-4, D-1
done
clear
View Answer play_arrow
Which one of the following is not correct?
A)
In case of malic acid, amount of \[C{{O}_{2}}\] released is more than \[{{O}_{2}}\] consumed and thus value of RQ is more than 1.
done
clear
B)
Cork cells are dead cells and hence do not respire.
done
clear
C)
In glycolysis, 4 ATP molecules are produced and two are used, hence net gain of ATP is two through direct synthesis.
done
clear
D)
Photorespiration is a useful phenomenon.
done
clear
View Answer play_arrow
Active uptake of minerals by roots mainly depends on the
A)
light
done
clear
B)
temperature
done
clear
C)
humidity
done
clear
D)
availability of oxygen
done
clear
View Answer play_arrow
Dentist's nerve is
A)
VIIth cranial nerve
done
clear
B)
IInd cranial nerve
done
clear
C)
Xth cranial nerve
done
clear
D)
Vth cranial nerve
done
clear
View Answer play_arrow
Glucose phosphates formed in photosynthesis are Asymmetrically labelled it is called
A)
Warburg's effect
done
clear
B)
Pasteur's effect
done
clear
C)
Dicken's effect
done
clear
D)
Gibb's effect
done
clear
View Answer play_arrow
Which of the following characteristic does not occur in Pinus?
A)
Pollen grains are produced in such a large number as to form yellowish cloud
done
clear
B)
Each vascular bundle in the long shoot of Pinus consists of xylem facing towards the centre of the shoot.
done
clear
C)
Pinus is a homos porous gymnosperm.
done
clear
D)
Microsporophyll of Pinus bears two oblong microsporangia abaxially on the proximal part.
done
clear
View Answer play_arrow
Which of the following is associated with\[{{C}_{2}}-cycle\]?
A)
Respiration
done
clear
B)
Photorespiration
done
clear
C)
Dark reaction of photosynthesis
done
clear
D)
Conversion of fat into sugar
done
clear
View Answer play_arrow
The original ration \[9:3:3:1\] ratio become modified into what ratio in dominant epistasis?
A)
\[9:3:4~\]
done
clear
B)
\[12:3:1~\]
done
clear
C)
\[13:3\]
done
clear
D)
\[9:7\]
done
clear
View Answer play_arrow
Which among the following is largest chromosome?
A)
X-chromosome
done
clear
B)
Polytene chromosome
done
clear
C)
Supernumery chromosome
done
clear
D)
Lamp brush chromosome
done
clear
View Answer play_arrow
Debove's membrane is a layer of
A)
epithelial tissue
done
clear
B)
connective tissue
done
clear
C)
muscular tissue
done
clear
D)
All of these
done
clear
View Answer play_arrow
Epiphytes like vanda develop special layer of absorptive tissue velamen consisting of 4 or 5 layers of long polygonal cells. Velamen takes part in
A)
absorption of water from soil
done
clear
B)
absorption of moisture from air
done
clear
C)
exchange of gases
done
clear
D)
transpiration
done
clear
View Answer play_arrow
Which of the following are subunit of 80S ribosome?
A)
60S and 20S
done
clear
B)
50S and 40S
done
clear
C)
60S and 40S
done
clear
D)
60S and 50S
done
clear
View Answer play_arrow
Which of the following is the basis of biological concept of species?
A)
Morphological feature
done
clear
B)
Method of reproduction
done
clear
C)
Morphology and method of reproduction
done
clear
D)
Reproductive isolation
done
clear
View Answer play_arrow
Little's leaf disease of brinjal takes place due to which one of the following reason?
A)
Copper
done
clear
B)
Molybdenum
done
clear
C)
Manganese
done
clear
D)
MLO
done
clear
View Answer play_arrow
First reaction in photosynthesis is
A)
photolysis of water
done
clear
B)
formation of \[NADP{{H}_{2}}\]
done
clear
C)
formation of ATP
done
clear
D)
excitation of chlorophyll molecule
done
clear
View Answer play_arrow
Silent valley National Park is located in
A)
Himalayas
done
clear
B)
Kerala
done
clear
C)
Nilgiri Hills
done
clear
D)
Tamil Nadu
done
clear
View Answer play_arrow
Select the correct statements. I. Pneumatophores are seen in Rhizophora II. Adventitious aerial roots are seen in banyan tree. III. In tridax the stem is decumbent. IV. If the stem is jointed with solid nodes and hollow in temodes, it is called caudex. V. Maize and sugarcane have prop roots.
A)
I, II and III
done
clear
B)
I, III and IV
done
clear
C)
II, IV and V
done
clear
D)
II, IV and III
done
clear
View Answer play_arrow
Abnormal development of spinal cord is
A)
myelodysplasia
done
clear
B)
tabes dorsalis
done
clear
C)
syringomyelia
done
clear
D)
neuritis
done
clear
View Answer play_arrow
Which of the following show molecular homology?
A)
Proteins in humans blood and blood of insects
done
clear
B)
Mouth parts of cockroach, honeybee, mosquito
done
clear
C)
Proteins in blood of man and ape
done
clear
D)
Fats in body of man and monkey
done
clear
View Answer play_arrow
During the DNA replication, the term leading strand is applied to which of the following strand
A)
strand replicating in \[3'\to 5'\] direction continuously
done
clear
B)
strand replicating in \[3'\to 5'\] direction discontinuously
done
clear
C)
strand replicating in \[5'\to 3'\] direction discontinuously
done
clear
D)
strand replicating in \[5'\to 3'\] direction continuously
done
clear
View Answer play_arrow
Vegetative propagation in mint and Chrysanthemum occurs by
A)
rhizome
done
clear
B)
sucker
done
clear
C)
runner
done
clear
D)
stolen
done
clear
View Answer play_arrow
Super Bug - a biotechnological achievement is useful in
A)
treatment of cancer
done
clear
B)
espionage
done
clear
C)
biodegradation
done
clear
D)
bioremediation of oil spills
done
clear
View Answer play_arrow
Which of the following is not an example of secondary structure of protein?
A)
Fibroin of silk
done
clear
B)
Ribonuclease
done
clear
C)
Haemoglobin
done
clear
D)
Hair keratin
done
clear
View Answer play_arrow
Which of the following component of blood does not enter into the nephron?
A)
Water
done
clear
B)
Urea
done
clear
C)
Plasma protein
done
clear
D)
Glucose
done
clear
View Answer play_arrow
Which among the following is 'prinbow box'?
A)
\[5'\,TAATTAG'\,3\]
done
clear
B)
\[3'\,TAATTAG'\,5\]
done
clear
C)
\[5' TATATTAG 3'\]
done
clear
D)
\[5' TATAATG 3'\]
done
clear
View Answer play_arrow
Which of the following is correctly matched?
A)
Wings of honeybee and crow - Homologous
done
clear
B)
Thorn of Bougainvillea and tendrils of Cucurbita - Analogous
done
clear
C)
Nephhdia of earthworm and malpighean tubules of cockroach - Excretory
done
clear
D)
Nictitating membrane and blind spot in human eye - Vestigial
done
clear
View Answer play_arrow
Which one of the following was observed for the first time by Trenb?
A)
Entry of pollen tube into the ovule through the micropyle in Casuarina
done
clear
B)
Entry of pollen tube into ovule through the chalaza in Casuarina
done
clear
C)
Entry of pollen tube into the ovule through the integuments in Hibiscus.
done
clear
D)
Entry of pollen tube into the ovule through the chalaza in Hibiscus.
done
clear
View Answer play_arrow
Vector is transmitting agant & organism of a disease. In case of filariasis, it is spread by
A)
sandfly
done
clear
B)
tse-tse fly
done
clear
C)
rat flea
done
clear
D)
Culex
done
clear
View Answer play_arrow
Scientific name of Java men is
A)
Homo rhodesiensis
done
clear
B)
Homo erectus
done
clear
C)
Pithecanthropus erectus
done
clear
D)
Sinanthropus pekinensis
done
clear
View Answer play_arrow
Phylloclade is photosynthetic, succulent specialized plant part found in
A)
Asparagus
done
clear
B)
Opuntia
done
clear
C)
Ulium
done
clear
D)
Euphorbia
done
clear
View Answer play_arrow
Eye of the moll us can group that resembles vertebrate eyes are
A)
pelecypoda
done
clear
B)
scaphopoda
done
clear
C)
cephalopoda
done
clear
D)
gastropoda
done
clear
View Answer play_arrow
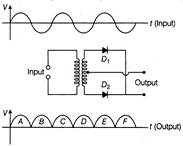 The contribution from the diode \[{{D}_{2}}\] is/are
The contribution from the diode \[{{D}_{2}}\] is/are 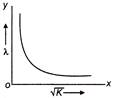
 is used as
is used as