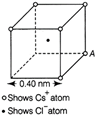
| (i) What is the net electric field on the C1 atom due to eight Cs atoms? |
| (ii) Suppose that the Cs atom at the corner A is missing. What is the net force now on the C1 atom due to seven remaining Cs atoms? |
Answer:
(i) From the given figure, we can analyse that the chlorine atom is at the centre of the cube, i.e. at equal distance from all the eight corners of cube where caesium atoms are placed. Thus, due to symmetry, the force due to all Cs atoms, on CI atom will cancel out. Since, \[E=F/q'\] \[[\text{where},F=0]\] \[\therefore \] E = 0 (ii) The force on CI atom at due to a single Cs atom would be given by \[F=\frac{{{e}^{2}}}{4\pi {{\varepsilon }_{0}}{{r}^{2}}}\] where, r = distance between CI ion and Cs ion. Applying Pythagorous theorem, we get \[r=\sqrt{{{(0.20)}^{2}}+{{(0.20)}^{2}}+{{(0.20)}^{2}}}\times {{10}^{-9}}m\] \[=0.346\times {{10}^{-9}}m\] Now,\[F=\frac{q}{4\pi {{\varepsilon }_{0}}{{r}^{2}}}=\frac{{{e}^{2}}}{4\pi {{\varepsilon }_{0}}{{r}^{2}}}=\frac{9\times {{10}^{9}}{{(1.6\times {{10}^{-19}})}^{2}}}{{{(0.346\times {{10}^{-9}})}^{2}}}\] \[=1.92\times {{10}^{-9}}N\] This is the net force on the CI-atom due to seven remaining Cs-atoms.
You need to login to perform this action.
You will be redirected in
3 sec
