Answer:
(a)
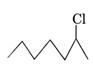 is a 2° alkyl
halide while
is a 2° alkyl
halide while ![]() is a 1° alkyl
halide.
Since 2° alkyl halide react faster than 1° alkyl halide due
to greater stability of 2° carbocation over 1° carbocation, therefore,
is a 1° alkyl
halide.
Since 2° alkyl halide react faster than 1° alkyl halide due
to greater stability of 2° carbocation over 1° carbocation, therefore, 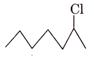 a is more faster
than,
a is more faster
than, 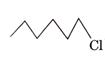 .
(b) Grignard reagents should be prepared under
anhydrous condition because the magnesium bond is covalent but highly polar
with carbon pulling electrons from electropositive magnesium, the
magnesium-halogen bond is essentially ionic. If water is present it convert
Grignard reagents to corresponding hydrocarbon. 1
.
(b) Grignard reagents should be prepared under
anhydrous condition because the magnesium bond is covalent but highly polar
with carbon pulling electrons from electropositive magnesium, the
magnesium-halogen bond is essentially ionic. If water is present it convert
Grignard reagents to corresponding hydrocarbon. 1
You need to login to perform this action.
You will be redirected in
3 sec
