Answer:
(a)
Haloarenes are less reactive towards nucleophilic substitution because the
benzene ring in haloarene is electron rich, also it withdraws
electrons from chlorine. Hence benzene becomes more rich in electrons and favor
electrophiles to attack it in comparison to nucleophilic attack is
easy. ![]()
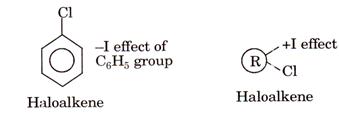
![]() (b)
(b) ![]() being a
stronger nucleophile than
being a
stronger nucleophile than ![]() , the
later cannot displace the former.
, the
later cannot displace the former.![]() protonates
ROH' to form
protonates
ROH' to form ![]() being
a weaker nucleophile than
being
a weaker nucleophile than ![]() , it is
easily displaced by latter. 1
, it is
easily displaced by latter. 1
You need to login to perform this action.
You will be redirected in
3 sec
