Answer:
Carbocation is the intermediate in the hydrolysis of each.
![]()
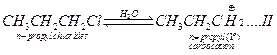 Allyl
carbocation is stabilised by resonance
Allyl
carbocation is stabilised by resonance
![]() This type of resonance is not in n-propyl carbocation. Thus
greater the stability, greater will be the formation. Hence, equilibrium
(I) is more spontaneous than (II), as a result hydrolysis of allyl chloride
is faster than n-propyl chloride. [2]
This type of resonance is not in n-propyl carbocation. Thus
greater the stability, greater will be the formation. Hence, equilibrium
(I) is more spontaneous than (II), as a result hydrolysis of allyl chloride
is faster than n-propyl chloride. [2]
You need to login to perform this action.
You will be redirected in
3 sec
