Answer:
Displacement
of copper from its' solution by iron can prove this.
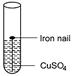
Activity
Place
iron nail in the copper sulphate solution taken in a test tube. After sometime
brown coloured copper metal is deposited on iron nail and colour of solution
slowly changes to pale green due to the formation of FeSC4 solution.
This proves
that iron is more reactive than copper.
![]()
 (1)
(1)
You need to login to perform this action.
You will be redirected in
3 sec
