Answer:
Burn a piece of soft coke in hard glass test tube and pass the gas produced in water. 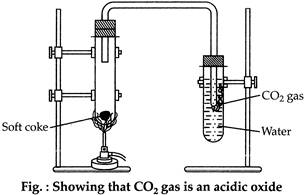
Then add blue litmus solution (to test the formation of acid) to the test-tube in which carbon dioxide gas is dissolved. The colour of the solution turns red. The following chemical reactions are going on during this experiment. (i) \[\begin{align} & C+{{O}_{2}}\to C{{O}_{2}} \\ & \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(Carbon\,\,dioxide) \\ \end{align}\] (ii) \[\begin{align} & C{{O}_{2}}+{{H}_{2}}O\to {{H}_{2}}C{{O}_{3}} \\ & \,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,\,(Carbonic\,\,acid) \\ \end{align}\]
You need to login to perform this action.
You will be redirected in
3 sec
