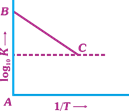
| Figure shows a graph in \[{{\log }_{10}}\,k\,vs\,\frac{1}{T}\] where, k is rate constant and T is temperature. The straight line BC has slope, \[\tan \,\theta \,=-\frac{1}{2.303}\] and an intercept of 5 on y-axis. Thus, \[{{E}_{a}},\] the energy of activation, is |
 |
A) 4.606 cal
B) \[\frac{0.2}{2.303}cal\]
C) 2 cal
D) None of these
Correct Answer: C
Solution :
| Rate constant, \[k=A{{e}^{-{{E}_{a}}/RT}}\] |
| In \[k=\frac{-{{E}_{a}}}{RT}+\ln \,A\] |
| \[2.303{{\log }_{10}}k=\frac{-{{E}_{a}}}{RT}+2.303{{\log }_{10}}A'\] |
| \[{{\log }_{10}}k=\frac{-{{E}_{a}}}{2.303R}\cdot \frac{1}{T}+{{\log }_{10}}A'\] |
| Now, \[\frac{-{{E}_{a}}}{2.303R}=\tan \theta \,=-\frac{1}{2.303}\] |
| \[\therefore \] \[{{E}_{a}}=R=2\,cal\] |
You need to login to perform this action.
You will be redirected in
3 sec
