A) \[\frac{13R}{6}\]
B) \[\frac{19R}{6}\]
C) \[\frac{23R}{6}\]
D) \[\frac{17R}{6}\]
Correct Answer: B
Solution :
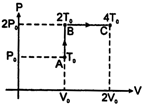
| [b]Let initial pressure, volume, temperature be \[{{P}_{0}},{{V}_{0}},{{T}_{0}}\] indicated by state A in P-V diagram. The gas is then isochoric ally taken to state \[B(2{{P}_{0}},{{V}_{0}},2{{T}_{0}})\] and then taken from state B to state \[C(2{{P}_{0}},2{{V}_{0}},4{{T}_{0}})\] isobaric ally. Total heat absorbed by 1 mole of gas |
| \[\Delta Q={{C}_{v}}(2{{T}_{0}}-{{T}_{0}})+{{C}_{P}}(4{{T}_{0}}-2{{T}_{0}})=\frac{5}{2}\] |
| \[R{{T}_{0}}+\frac{7}{2}R\times 2{{T}_{0}}=\frac{19}{2}R{{T}_{0}}\] |
| Total change in temperature from state A to C is \[\Delta T=3{{T}_{0}}\] |
 |
| \[\therefore \] Molar heat capacity |
| \[=\frac{\Delta Q}{\Delta T}=\frac{\frac{19}{2}R{{T}_{0}}}{3{{T}_{0}}}=\frac{19}{6}R.\] |
You need to login to perform this action.
You will be redirected in
3 sec
