A) Saytzeff rule
B) Markownikoff's rule
C) Kharasch effect
D) Anti-Saytzeff rule
Correct Answer: B
Solution :
The Markownikoffs rule : During the addition across unsymmetrical double bond the negative part of the adding molecule attaches itself to the carbon atom Carrying less number of hydrogen atoms.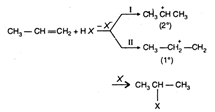 As \[{{\text{3}}^{\text{o}}}\] and \[{{2}^{\text{o}}}\] carbocation are more stable than \[{{1}^{\text{o}}}\] carbocation hence the addition through path I is favoured which is in accordance with Markownikoffs rule. Kharasch effect or peroxide effect: In the presence of organic peroxides such as benzoyl peroxide (addition on alkenes give the product against the Markownikoffs rule. \[C{{H}_{3}}CH=C{{H}_{2}}+HBr\xrightarrow{Peroxide}C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Br\] Saytzeff rule: During dehydrohalogenation of alkyl halide, the hydrogen atom is removed from that carbon atom which has lesser number of carbon atoms and more number of alkyi groups attached to it i.e., to form more alkylated alkene
As \[{{\text{3}}^{\text{o}}}\] and \[{{2}^{\text{o}}}\] carbocation are more stable than \[{{1}^{\text{o}}}\] carbocation hence the addition through path I is favoured which is in accordance with Markownikoffs rule. Kharasch effect or peroxide effect: In the presence of organic peroxides such as benzoyl peroxide (addition on alkenes give the product against the Markownikoffs rule. \[C{{H}_{3}}CH=C{{H}_{2}}+HBr\xrightarrow{Peroxide}C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}Br\] Saytzeff rule: During dehydrohalogenation of alkyl halide, the hydrogen atom is removed from that carbon atom which has lesser number of carbon atoms and more number of alkyi groups attached to it i.e., to form more alkylated alkene 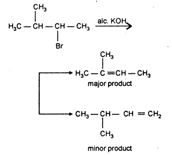
You need to login to perform this action.
You will be redirected in
3 sec
