A) formaldehyde
B) ethanol
C) glyoxal
D) propanaldehyde
Correct Answer: C
Solution :
\[\text{C}{{\text{H}}_{2}}=CH-CH=CH-C{{H}_{3}}\xrightarrow{{{O}_{3}}}\]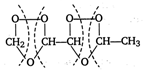 \[\xrightarrow{Zn/{{H}_{2}}O}\underset{\text{formaldehyde}}{\mathop{HCHO}}\,+\underset{\begin{smallmatrix} CHO \\ \text{glyoxal} \end{smallmatrix}}{\mathop{\underset{|}{\mathop{C}}\,}}\,HO+\underset{\text{acetaldehyde}}{\mathop{C{{H}_{3}}CHO}}\,\]
\[\xrightarrow{Zn/{{H}_{2}}O}\underset{\text{formaldehyde}}{\mathop{HCHO}}\,+\underset{\begin{smallmatrix} CHO \\ \text{glyoxal} \end{smallmatrix}}{\mathop{\underset{|}{\mathop{C}}\,}}\,HO+\underset{\text{acetaldehyde}}{\mathop{C{{H}_{3}}CHO}}\,\]
You need to login to perform this action.
You will be redirected in
3 sec
