A) +54kJ
B) +219kJ
C) -219kJ
D) -165kJ
Correct Answer: D
Solution :
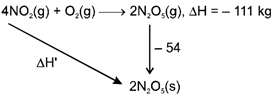 \[-111-54=\Delta H'\] \[\Delta H'=-165KJ\]
\[-111-54=\Delta H'\] \[\Delta H'=-165KJ\]
You need to login to perform this action.
You will be redirected in
3 sec
