A) (i) > (ii) > (iii) >(iv)
B) (ii) > (iv) > (iii) > (i)
C) (ii) > (iv) > (i) > (iii)
D) (ii), > (iii) > (iv) > (i)
Correct Answer: D
Solution :
(i)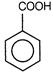 (ii)
(ii) 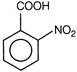 (iii)
(iii) 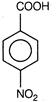 (iv)
(iv) 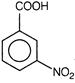
You need to login to perform this action.
You will be redirected in
3 sec
