A) \[{{d}^{5}}\](in strong ligand field)
B) \[{{d}^{3}}\](in weak as well as in strong fields)
C) \[{{d}^{4}}\](in weak ligand field)
D) \[{{d}^{4}}\](in strong ligand field)
Correct Answer: D
Solution :
Magnetic moments depends on the number of unpaired electrons. Pairing of electrons in complex depends on the ligand filled strength. Therefore, calculate the magnetic moment for different configuration given in question. \[{{d}^{5}}\]in strong field \[n=\]unpaired electron\[=1\]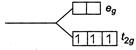 \[n=3\] Magnetic moment \[=\sqrt{15}=3.87\,BM\] \[{{d}^{4}}\]in weak field \[n=4\]
\[n=3\] Magnetic moment \[=\sqrt{15}=3.87\,BM\] \[{{d}^{4}}\]in weak field \[n=4\] 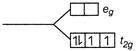 \[n=2\] Magnetic moment\[=\sqrt{8}=2.83\text{ }BM\]
\[n=2\] Magnetic moment\[=\sqrt{8}=2.83\text{ }BM\]
You need to login to perform this action.
You will be redirected in
3 sec
