A) one, tetrahedral
B) two, tetrahedral
C) one, square planar
D) two, square planar
Correct Answer: B
Solution :
\[_{28}Ni=[Ar]3{{s}^{2}}3{{p}^{6}}4{{s}^{2}}3{{d}^{8}}\] \[N{{i}^{2+}}=[Ar]3{{s}^{2}}3{{p}^{6}}3{{d}^{6}}\]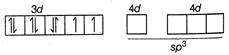 Nickel has two unpaired electrons and geometry is tetrahedral due to\[s{{p}^{3}}\]hybridisation.
Nickel has two unpaired electrons and geometry is tetrahedral due to\[s{{p}^{3}}\]hybridisation.
You need to login to perform this action.
You will be redirected in
3 sec
