A) smaller size of B - atom as compared to that of C - atom
B) stronger\[\sigma \]bond between B and F in\[B{{F}_{3}}\]as compared to that between C and F in\[C{{F}_{4}}\]
C) significant\[p\pi -p\pi \]interaction between B and F in\[B{{F}_{3}}\]whreas there is no possibility of such interaction between C and F in\[C{{F}_{4}}\]
D) lower degree of \[p\pi -p\pi \] interaction between B and F in\[B{{F}_{3}}\]than that between C and F in\[C{{F}_{4}}\]
Correct Answer: C
Solution :
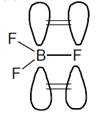 B has vacant available p orbital and hence it involves\[p\pi -p\pi \]back bonding which is not possible in\[C{{F}_{4}}\]as C does not have vacant orbital.
B has vacant available p orbital and hence it involves\[p\pi -p\pi \]back bonding which is not possible in\[C{{F}_{4}}\]as C does not have vacant orbital.
You need to login to perform this action.
You will be redirected in
3 sec
