A) \[C{{H}_{3}}COC{{H}_{2}}COOH\]
B) \[C{{H}_{3}}COC{{H}_{3}}\]
C)
![]()
D)
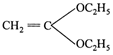
Correct Answer: A
Solution :
The reaction is known as Claisen condensation \[C{{H}_{3}}COC{{H}_{2}}COO{{C}_{2}}{{H}_{5}}\xrightarrow{(i)\,\,{{C}_{2}}{{H}_{5}}ONa}\]\[\underset{\underset{(A)}{\mathop{Ethyl\,acetoaacetate}}\,}{\mathop{C{{H}_{3}}COC{{H}_{2}}COO{{C}_{2}}{{H}_{5}}}}\,+\underset{Ethanol}{\mathop{{{C}_{2}}{{H}_{5}}OH}}\,\] \[\underset{(A)}{\mathop{C{{H}_{3}}COC{{H}_{2}}COO{{C}_{2}}{{H}_{5}}}}\,\xrightarrow{{{H}_{3}}{{O}^{+}}}\] \[\underset{(B)}{\mathop{C{{H}_{3}}COC{{H}_{2}}COOH}}\,+{{C}_{2}}{{H}_{5}}OH\]You need to login to perform this action.
You will be redirected in
3 sec
