Direction for: In each of the following questions a statement of Assertion is given followed by a corresponding statement of Reason just below it. Of the statements mark the correct answer as
Assertion: \[{{K}_{4}}[Fe{{(CN)}_{6}}]\] is diamagnetic and \[[Fe{{({{H}_{2}}O)}_{6}}]C{{l}_{3}}\] is paramagnetic. Reason: Hybridisation of central metal in \[{{K}_{4}}[Fe{{(CN)}_{6}}]\] is \[s{{p}^{3}}{{d}^{2}},\] while in \[[Fe{{({{H}_{2}}O)}_{6}}]C{{l}_{3}}\]is \[{{d}^{2}}s{{p}^{3}}\].A) If both Assertion: and Reason: are true and the Reason: is the correct explanation of the Assertion:
B) If both Assertion: and Reason: are true but the Reason: is not the correct explanation of the Assertion:
C) If Assertion: is true but Reason: is false
D) If both Assertion: and Reason: are false
Correct Answer: C
Solution :
In \[(iv)<(ii)<(iii)<(i)\], Fe is present as \[(i)<(iii)<(iv)<(ii)\]. \[{{[Cr{{(en)}_{3}}]}^{3+}}\]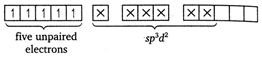 Since, it contains five unpaired electrons, it is paramagnetic in nature.
Since, it contains five unpaired electrons, it is paramagnetic in nature.
You need to login to perform this action.
You will be redirected in
3 sec
