A) \[C{{H}_{3}}C{{H}_{2}}C{{H}_{2}}C{{H}_{2}}COO{{C}_{2}}{{H}_{5}}\]
B) \[{{C}_{6}}{{H}_{5}}COO{{C}_{2}}{{H}_{5}}\]
C) \[{{C}_{6}}{{H}_{5}}C{{H}_{2}}COO{{C}_{2}}{{H}_{5}}\]
D) \[{{C}_{6}}{{H}_{11}}C{{H}_{2}}COO{{C}_{2}}{{H}_{5}}\]
Correct Answer: B
Solution :
It is due to presence of \[\alpha \]-hydrogen atom in ester \[C{{H}_{3}}-C{{H}_{2}}-C{{H}_{2}}-C{{H}_{2}}-\]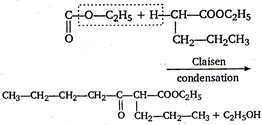 \[{{C}_{6}}{{H}_{5}}COO{{C}_{2}}{{H}_{5}}+{{C}_{6}}{{H}_{5}}COO{{C}_{2}}{{H}_{5}}\] \[\to \] No reaction because for Claisen condensation an ester with two \[\alpha \]-hydrogens atoms are required
\[{{C}_{6}}{{H}_{5}}COO{{C}_{2}}{{H}_{5}}+{{C}_{6}}{{H}_{5}}COO{{C}_{2}}{{H}_{5}}\] \[\to \] No reaction because for Claisen condensation an ester with two \[\alpha \]-hydrogens atoms are required 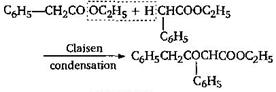
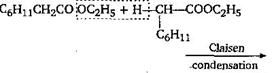 \[{{C}_{6}}{{H}_{11}}\,C{{H}_{2}}CO\underset{\begin{smallmatrix} | \\ {{C}_{6}}{{H}_{11}} \end{smallmatrix}}{\mathop{C}}\,H-COO{{C}_{2}}{{H}_{5}}+{{C}_{2}}{{H}_{5}}OH\]
\[{{C}_{6}}{{H}_{11}}\,C{{H}_{2}}CO\underset{\begin{smallmatrix} | \\ {{C}_{6}}{{H}_{11}} \end{smallmatrix}}{\mathop{C}}\,H-COO{{C}_{2}}{{H}_{5}}+{{C}_{2}}{{H}_{5}}OH\]
You need to login to perform this action.
You will be redirected in
3 sec
