A)
![]()
B)
![]()
C)
![]()
D)
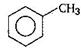
Correct Answer: C
Solution :
?CI is able to deactivate the benzene nucleus but show o/p-directive influence while ?OH and \[-C{{H}_{3}}\] activate the benzene nucleus (order \[-OH>-C{{H}_{3}}\]) so, they show electrophilic substitution easily on o/p position in comparison to benzene. Order of electrophilic substitution Thus phenol will be most easily attacked by an electrophile. Order of electrophilic substitution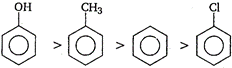
You need to login to perform this action.
You will be redirected in
3 sec
