| List-I | List-II |
| A. Benzaldehyde | 1. Phenolphthalein |
| B. Phthalic anhydride | 2. Benzoin condensation |
| C. Phenyl benzoate | 3. Oil wintergreen |
| D. Methyl salicylate | 4. Fries rearrangement |
A) A-4 B-1 C-3 D-2
B) A-4 B-2 C-3 D-1
C) A-2 B-3 C-4 D-1
D) A-2 B-1 C-4 D-3
Correct Answer: D
Solution :
[a] Benzoin condensation. When an ethanolic solution of benzaldehyde is heated with strong alkali like KCN or NaCN, we get benzoin. \[\text{2}{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}-\overset{\begin{smallmatrix} \text{O} \\ \text{ }\!\!|\!\!\text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{C}}}\,-\text{H}\xrightarrow[{}]{\text{C}{{\text{N}}^{\text{ }\!\!\Theta\!\!\text{ }}}}{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}\overset{\begin{smallmatrix} \text{O} \\ \text{ }\!\!|\!\!\text{ }\!\!|\!\!\text{ } \end{smallmatrix}}{\mathop{\text{C}}}\,-\underset{\text{benzoin}}{\mathop{\underset{\begin{smallmatrix} \text{ }\!\!|\!\!\text{ } \\ \text{OH} \end{smallmatrix}}{\mathop{\text{CH}}}\,}}\,-{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{5}}}\] [b] Formation of phenolphthalein. When phenol is treated with phthalic anhydride in the presence of concentrated \[{{\text{H}}_{\text{2}}}\text{S}{{\text{O}}_{\text{4}}}\text{,}\]it gives phenolphthalein, an indicator.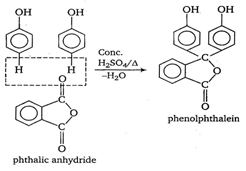 [c] Fries rearrangement Phenyl benzoate when heated with anhydrous \[AlC{{l}_{3}}\] in the presence of inert solvent gives ortho-and para-hydroxybenzophenone. In this rearrangement, there is a benzoyl group migration from the phenolic oxygen to an ortho-and para-position.
[c] Fries rearrangement Phenyl benzoate when heated with anhydrous \[AlC{{l}_{3}}\] in the presence of inert solvent gives ortho-and para-hydroxybenzophenone. In this rearrangement, there is a benzoyl group migration from the phenolic oxygen to an ortho-and para-position. 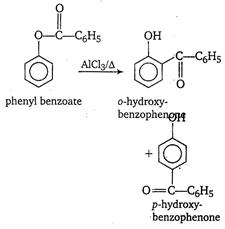 (A chief constituent of oil of wintergreen)
(A chief constituent of oil of wintergreen)
You need to login to perform this action.
You will be redirected in
3 sec
