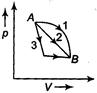
A) \[{{Q}_{1}}>{{Q}_{2}}>{{Q}_{3}}and\Delta {{U}_{1}}=\Delta {{U}_{2}}=\Delta {{U}_{3}}\]
B) \[{{Q}_{3}}>{{Q}_{2}}>{{Q}_{1}}and\Delta {{U}_{1}}=\Delta {{U}_{2}}=\Delta {{U}_{3}}\]
C) \[{{Q}_{1}}={{Q}_{2}}={{Q}_{3}}and\Delta {{U}_{1}}>\Delta {{U}_{2}}>\Delta {{U}_{3}}\]
D) \[{{Q}_{3}}>{{Q}_{2}}>{{Q}_{1}}and\Delta {{U}_{1}}>\Delta {{U}_{2}}>\Delta {{U}_{3}}\]
Correct Answer: A
Solution :
For all process 1, 2 and 3 \[\Delta U={{U}_{B}}-{{U}_{A}}\]is same \[\therefore \] \[\Delta {{U}_{1}}=\Delta {{U}_{2}}=\Delta {{U}_{3}}\] Now, \[\Delta Q=\Delta U+\Delta W\] Now, \[\Delta W=\] work done by the gas \[\therefore \] \[\Delta {{Q}_{1}}=\Delta {{Q}_{2}}>\Delta {{Q}_{3}}\]You need to login to perform this action.
You will be redirected in
3 sec
