A) (T + 2.4) K
B) (T - 2.4) K
C) (T + 4) K
D) (T - 4) K
Correct Answer: D
Solution :
Key Idea: in an adiabatic process, there is no heat transfer into or out of a system \[i.e.,\,Q=0\] In an adiabatic process \[Q=0\] So, from 1st law of thermodynamics. \[W=-\Delta U\] \[=-n{{C}_{V}}\Delta T\]\[=-\left( \frac{R}{\gamma -1} \right)({{T}_{f}}-{{T}_{i}})\]\[=\frac{nR}{\gamma -1}({{T}_{i}}-{{T}_{f}})\] Here: \[W=6R\,\,\,J,\,\,n=1\,\,\,mol,\] \[R=8.31\,J/mol-K,\,\,\gamma =\frac{5}{3},{{T}_{i}}=T\,K\] Substituting given values in Eq. (i), we get \[\therefore \]\[6R=\frac{R}{(5/3-1)}(T-{{T}_{f}})\] \[\Rightarrow \]\[6R=\frac{3R}{2}(T-{{T}_{f}})\] \[\Rightarrow \]\[T-{{T}_{f}}=4\] \[\therefore \]\[{{T}_{f}}=(T-4)K\] NOTE: Adiabatic expansions of mono, dia and polyatomic gases are shown below 1\[\to \]monoatomic 2\[\to \] diatomic 3\[\to \] polyatomic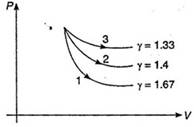
You need to login to perform this action.
You will be redirected in
3 sec
