A) \[{{\text{H}}_{\text{3}}}\text{C}\text{C}{{\text{H}}_{\text{2}}}\text{OH}+\text{ }{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}\]
B) \[\text{C}{{\text{H}}_{\text{3}}}\text{CH}=\text{C}{{\text{H}}_{\text{2}}}+{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}\]
C) \[{{\text{H}}_{\text{2}}}\text{C}=\text{C}{{\text{H}}_{\text{2}}}\text{+}{{\text{C}}_{\text{6}}}{{\text{H}}_{6}}\]
D) \[{{\text{H}}_{\text{3}}}\text{C}-\text{C}{{\text{H}}_{\text{3}}}+{{\text{C}}_{\text{6}}}{{\text{H}}_{\text{6}}}\]
Correct Answer: C
Solution :
By the reaction of benzene with ethylene in presence of anhydrous. \[\text{A1C}{{\text{1}}_{\text{3}}},\]ethyl benzene is produced,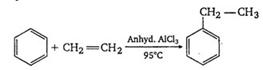
You need to login to perform this action.
You will be redirected in
3 sec
