A)
![]()
B)
![]()
C)
![]()
D)
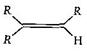
Correct Answer: A
Solution :
Stability of alkene: \[\propto \,\,\frac{1}{Heat\,of\,hydrogenation\,of\,alkene}\] Greater the number of alkyl groups attached to the doubly bonded carbon atoms, more stable is the alkene. Hence, given alkene follow the following order of stability.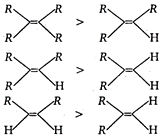 Hence, faster hydrogenation in
Hence, faster hydrogenation in You need to login to perform this action.
You will be redirected in
3 sec
